Summary
Forskolin (i.e, cAMP)-modulation of ion transport pathways in filter-grown monolayers of the Cl−-secreting subclone (19A) of the human colon carcinoma cell line HT29 was studied by combined Ussing chamber and microimpalement experiments.
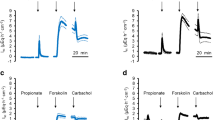
Changes in electrophysiological parameters provoked by serosal addition of 10−5 m forskolin included: (i) a sustained increase in the transepithelial potential difference (3.9±0.4 mV). (ii) a transient decrease in transepithelial resistance with 26±3 Ω · cm2 from a mean value of 138±13 Ω · cm2 before forskolin addition, (iii) a depolarization of the cell membrane potential by 24±1 mV from a resting value of −50±1 mV and (iv) a decrease in the fractional resistance of the apical membrane from 0.80±0.02 to 0.22±0.01. Both, the changes in cell potential and the fractional resistance, persisted for at least 10 min and were dependent on the presence of Cl− in the medium. Subsequent addition of bumetanide (10−4 m), an inhibitor of Na/K/2Cl cotransport, reduced the transepithelial potential, induced a repolarization of the cell potential and provoked a small increase of the transepithelial resistance and fractional apical resistance. Serosal Ba2+ (1mm), a known inhibitor of basolateral K+ conductance, strongly reduced the electrical effects of forskolin. No evidence was found for a forskolin (cAMP)-induced modulation of basolateral K+ conductance.
The results suggest that forskolin-induced Cl− secretion in the HT-29 cl.19A colonic cell line results mainly from a cAMP-provoked increase in the Cl− conductance of the apical membrane but does not affect K+ or Cl− conductance pathways at the basolateral pole of the cell. The sustained potential changes indicate that the capacity of the basolateral transport mechanism for Cl− and the basal Ba2+-sensitive K+ conductance are sufficiently large to maintain the Cl− efflux across the apical membrane. Furthermore, evidence is presented for an anomalous inhibitory action of the putative Cl− channel blockers NPPB and DPC on basolateral conductance rather than apical Cl− conductance.
Similar content being viewed by others
References
Augeron, C., Laboisse, C.L. 1984. Emergence of permanently differentiated cell clones in a human colonic cancer cell line in culture after treatment with sodium butyrate.Cancer Res. 44:3961–3969
Augeron, C., Maoret, J.J., Laboisse, C.L., Grasset, E. 1986. Permanently differentiated cell clones isolated from the human colonic adenocarcinoma cell line HT29: Possible models for the study of ion transport and mucus production.In: Ion Gradient Coupled Transport. F. Alvarado and C.H. van Os, editors, pp. 363–366. Elsevier, Amsterdam
Augeron, C., Popowicz, P., Simmons, N.L. 1987. Bumetanide inhibition of ouabain-insensitive86Rb(K) transport in a cultured human colonic adenocarcinoma cell-line (HT29 cl.19A)J. Physiol. 391:100P (Abstr.)
Bajnath, R., Augeron, C., Maoret, J.J., Laboisse, C., de Jonge, H.R., Groot, J.A. 1989. Comparison of electrophysiological changes induced by carbachol in T84 and HT-29 cl.19A human colon carcinoma cell lines.Z. Gastroenterol. 27:287
Bakker, R., Groot, J.A. 1989. Further evidence for the regulation of the tight junction ion selectivity by cAMP in goldfish intestinal mucosa.J. Membrane Biol. 111:25–35
Burnham, D.B., Fondacaro, J.D. 1989. Secretagogue-induced protein phosphorylation and chloride transport in Caco-2 cells.Am. J. Physiol. 256:G808-G816
Cliff, W.H., Frizzell, R.A. 1990. Separate Cl− conductances activated by cAMP and Ca2+ in Cl− secreting epithelial cells.Proc. Natl. Acad. Sci. USA 87:4956–4960
Dharmsathaphorn, K., Mandel, K.G., Masui, H., McRoberts, J.A. 1985. Vasoactive intestinal polypeptide-induced chloride secretion by a colonic epithelial cell line: Direct participation of a basolaterally localized Na,K,Cl cotransport system.J. Clin. Invest. 75:462–471
Diener, M., Rummel, W. 1989. Actions of the Cl− channelblocker NPPB on absorptive and secretory transport processes of Na+ and Cl− in rat descending colon.Acta Physiol. Scand. 137:215–222
Frizzell, R.A., Field, M., Schultz, S.G. 1979. Sodium-coupled chloride transport by epithelial tissues.Am. J. Physiol. 236:F1-F8
Grasset, E., Bernabeu, J., Pinto, M. 1985. Epithelial properties of human colonic carcinoma cell line Caco-2: Effect of secretagogues.Am. J. Physiol. 248:C410-C418
Grasset, E., Pinto, M., Dussaulx, E., Zweibaum, A., Desjeux, J.F. 1984. Epithelial properties of human colonic carcinoma cell line Caco-2: Electrical parameters.Am. J. Physiol. 247:C260-C267
Groot, J.A., Bakker, R. 1988. NaCl transport in the vertebrate intestine.In: Advances in Comparative and Environmental Physiology. R. Greger, editor. Vol. 1, pp. 103–152. Springer-Verlag, Berlin
Groot, J.A., Bakker, R., Bijlsma, P., Oosterhuis, W.P. 1987. The response of stripped rat ileum to forskolin is partially mediated by neuronal cells.Z. Gastroenterol. 45:409 (Abstr)
Hayslett, J.P., Gogelein, H., Kunzelman, K., Greger, R. 1987. Characteristics of apical chloride channels in human colon cells (HT29).Pfluegers Arch. 410:487–494
Heintze, K., Stewart, C.P., Frizzell, R.A. 1983. Sodium dependent chloride secretion across rabbit descending colon.Am. J. Physiol. 244:G357-G365
Horvath, P.J., Fernandez, P.C., Weiser, M., Duffey, M.E. 1986. Localization of chloride secretion in rabbit colon: Inhibition by anthracene-9-carboxylic acid.Am. J. Physiol. 250:G185-G190
Kunzelmann, K., Pavenstadt, H., Greger, R. 1989. Properties and regulation of chloride channels in cystic fibrosis and normal airway cells.Pfluegers Arch. 415:172–182
McCann, J.D., Bhalla, R.C., Welsh, M. 1989. Release of intracellular calcium by two different second messengers in airway epithelium.Am. J. Physiol. 257:L116-L124
McCann, J.D., Matsuda, J., Garcia, M., Kaczorowski, G., Welsh, M. 1990. Basolateral K+ channels in airway epithelia I. Regulation by Ca and block by charybdotoxin.Am. J. Physiol. 258:L334-L342
McRoberts, J.A., Beuerlein, G., Dharmsathaphorn, K. 1985. Cyclic AMP and Ca++ activated K+ transport in human colonic epithelial cell line.J. Biol. Chem. 260:14163–14172
Shorofsky, S.R., Field, M., Fozzard, H.A. 1983. Electrophysiology of Cl− secretion in canine trachea.J. Membrane Biol. 72:105–115
Stewart, C.P., Turnberg, L.A. 1989. A microelectrode study of responses to secretagogues by epithelial cells on villus and crypt of rat small intestine.Am. J. Physiol. 257:G334-G343
Wangemann, P., Di Stefano, A., Wittner, M., Englert, H.C., Lang, H.J., Schlatter, E., Greger, R. 1986. Cl− channel blockers in the thick ascending limb of the loop of Henle. Structure activity relationship.Pfluegers Arch. 407:S128-S141
Welsh, M.J. 1987. Electrolyte transport by airway epithelia.Physiol. Rev. 67:1143–1184
Welsh, M.J., Smith, P.L., Frizzell, R. 1983. Chloride secretion by canine tracheal epithelium: III. Membrane resistances and electromotive forces.J. Membrane Biol. 71:209–218
Welsh, M.J., Smith, P.L., Fromm, M., Frizzell, R.A. 1982. Crypts are the site of intestinal fluid and electrolyte secretion.Science 218:1219–1221
Willumsen, N.J., Davis, C.W., Boucher, R.C. 1989. Intracellular Cl− activity and cellular Cl− pathways in cultured human airway epithelium.Am. J. Physiol. 256:C1033-C1044
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bajnath, R.B., Augeron, C., Laboisse, C.L. et al. Electrophysiological studies of forskolin-induced changes in ion transport in the human colon carcinoma cell line HT-29 cl.19A: Lack of evidence for a cAMP-activated basolateral K+ conductance. J. Membrain Biol. 122, 239–250 (1991). https://doi.org/10.1007/BF01871424
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01871424