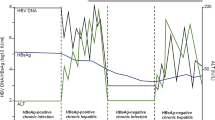
Abstract
Large volume of new data on the natural history and treatment of chronic hepatitis B virus (HBV) infection have become available since 2008. These include further studies in asymptomatic subjects with chronic HBV infection and community-based cohorts, the role of HBV genotype/naturally occurring HBV mutations, the application of non-invasive assessment of hepatic fibrosis and quantitation of HBV surface antigen and new drug or new strategies towards more effective therapy. To update HBV management guidelines, relevant new data were reviewed and assessed by experts from the region, and the significance of the reported findings was discussed and debated. The earlier “Asian-Pacific consensus statement on the management of chronic hepatitis B” was revised accordingly. The key terms used in the statement were also defined. The new guidelines include general management, indications for fibrosis assessment, time to start or stop drug therapy, choice of drug to initiate therapy, when and how to monitor the patients during and after stopping drug therapy. Recommendations on the therapy of patients in special circumstances, including women in childbearing age, patients with antiviral drug resistance, concurrent viral infection, hepatic decompensation, patients receiving immune suppression or chemotherapy and patients in the setting of liver transplantation and hepatocellular carcinoma, are also included.



Similar content being viewed by others
References
Liaw YF, Leung N, Kao JH, et al. Asian-Pacific consensus statement on the management of chronic hepatitis B: a 2008 update. Hepatol Int 2008;2:263–283
Keeffe EB, Dieterich DT, Han SH, et al. A treatment algorithm for the management of chronic hepatitis B virus infection in the United States: 2008 update. Clin Gastroenterol Hepatol 2008;6:1315–1341
Lok AS, McMahon BJ. Chronic hepatitis B: update 2009. Hepatology 2009;50:661–662
European Association For The Study Of The Liver. EASL clinical practice guidelines: management of chronic hepatitis B. J Hepatol 2009;50:227–242
Liaw YF. Antiviral therapy of chronic hepatitis B: opportunities and challenges in Asia. J Hepatol 2009;51:403–410
Liaw YF, Chu CM. Hepatitis B virus infection. Lancet 2009;373:582–592
Hui CK, Leung N, Yuen ST, et al. Natural history and disease progression in Chinese chronic hepatitis B patients in immune-tolerant phase. Hepatology 2007;46:395–401
Lai M, Hyatt BJ, Nasser I, et al. The clinical significance of persistently normal ALT in chronic hepatitis B infection. J Hepatol 2007;47:760–767
Liaw YF. Hepatitis flares and hepatitis B e antigen seroconversion: Implication in anti-hepatitis B virus therapy. J Gastroenterol Hepatol 2003;18:246–252
Lin CL, Kao JH. The clinical implications of hepatitis B virus genotype: recent advances. J Gastroenterol Hepatol 2011;26(Suppl 1):123–130
Chu CM, Hung SJ, Lin J, et al. Natural history of hepatitis B e antigen to antibody seroconversion in patients with normal serum aminotransferase levels. Am J Med 2004;116:829–834
Hsu YS, Chien RN, Yeh CT, et al. Long-term outcome after spontaneous HBeAg seroconversion in patients with chronic hepatitis B. Hepatology 2002;35:1522–1527
Chu CM, Liaw YF. Predictive factors for reactivation of hepatitis B following hepatitis B e antigen seroconversion in chronic hepatitis B. Gastroenterology 2007;133:1458–1465
Chen YC, Chu CM, Liaw YF. Age-specific prognosis following spontaneous hepatitis B e antigen seroconversion in chronic hepatitis B. Hepatology 2010;51:435–444
Chu CM, Liaw YF. Incidence and risk factors of progression to cirrhosis in inactive carriers of hepatitis B virus. Am J Gastroenterol 2009;104:1693–1699
Feld JJ, Ayers M, El-Ashry D, et al. Hepatitis B virus DNA prediction rules for hepatitis B e antigen-negative chronic hepatitis B. Hepatology 2007;46:1057–1070
Liaw YF, Tai DI, Chu CM, Chen TJ. The development of cirrhosis in patients with chronic type B hepatitis: a prospective study. Hepatology 1988;8:493–496
Lin SM, Yu ML, Lee CM, et al. Interferon therapy in HBeAg positive chronic hepatitis reduces cirrhosis and hepatocellular carcinoma. J Hepatol 2007;46:45–52
Park BK, Park YN, Ahn SH, et al. Long-term outcome of chronic hepatitis B based on histological grade and stage. J Gastroenterol Hepatol 2007;22:383–388
Wu CF, Yu MW, Lin CL, et al. Long-term tracking of hepatitis B viral load and the relationship with risk for hepatocellular carcinoma in men. Carcinogenesis 2008;29:106–112
Chen CF, Lee WC, Yang HI, et al. Changes in serum levels of HBV DNA and alanine aminotransferase determine risk for hepatocellular carcinoma. Gastroenterology 2011;141:1240–1248
Chen YC, Chu CM, Yeh CT, Liaw YF. Natural course following the onset of cirrhosis in patients with chronic hepatitis B: a long term follow-up study. Hepatol Int 2007;1:267–273
Chu CM, Liaw YF. Hepatitis B virus-related cirrhosis: natural history and treatment. Semin Liver Dis 2006;26:142–152
Yang HI, Lu SN, Liaw YF, et al. Hepatitis B e antigen and the risk of hepatocellular carcinoma. N Engl J Med 2002;347:168–174
Iloeje UH, Yang HI, Su J, et al. Predicting cirrhosis risk based on the level of circulating hepatitis B viral load. Gastroenterology 2006;130:678–686
Chen CJ, Yang HI, Su J, et al. Risk of hepatocellular carcinoma across a biological gradient of serum hepatitis B virus DNA level. JAMA 2006;295:65–73
Chen CL, Yang HI, Yang WS, et al. Metabolic factors and risk of hepatocellular carcinoma by chronic hepatitis B/C infection: a follow-up study in Taiwan. Gastroenterology 2008;135:111–121
Yu MW, Shih WL, Lin CL, et al. Body-mass index and progression of hepatitis B: a population-based cohort study in men. J Clin Oncol 2008;26:5576–5582
Tai DI, Lin SM, Sheen IS, et al. Long-term outcome of hepatitis B e antigen-negative hepatitis B surface antigen carriers in relation to changes of alanine aminotransferase levels over time. Hepatology 2009;49:1859–1867
Chu CM, Chen YC, Tai DI, Liaw YF. Level of hepatitis B virus DNA in inactive carriers with persistently normal levels of alanine aminotransferase. Clin Gastroenterol Hepatol 2010;8:535–540
Chen YC, Huang SF, Chu CM, Liaw YF. Serial HBV DNA levels in patients with persistently normal transaminase over 10 years following spontaneous HBeAg seroconversion. J Viral Hepat 2012;19:138–146
Chen JD, Yang HI, Iloeje UH, et al. Carriers of inactive hepatitis B virus are still at risk for hepatocellular carcinoma and liver-related death. Gastroenterology 2010;138:1747–1754
Chu CM, Liaw YF. Hepatitis B surface antigen seroclearance during chronic HBV infection. Antivir Ther 2010;15:133–143
Liu J, Yang HI, Lee MH, et al. Incidence and determinants of spontaneous hepatitis B surface antigen seroclearance: a community-based follow-up study. Gastroenterology 2010;139:474–482
Chu CM, Liaw YF. Prevalence of and risk factors for hepatitis B viremia after spontaneous hepatitis B surface antigen seroclearance in hepatitis B carrier. Clin Infect Dis 2012;54:88–90
Liaw YF. Clinical utility of hepatitis B surface antigen quantitation in patients with chronic hepatitis B: a review. Hepatology 2011;53:2121–2129
Chan HL, Thompson A, Martinot-Peignoux M, et al. Hepatitis B surface antigen quantification: Why and how to use it in 2011—A Core Group Report. J Hepatol 2011;55:1121–1131
Nguyen T, Thompson AJ, Bowden S, et al. Hepatitis B surface antigen levels during the natural history of chronic hepatitis B: a perspective on Asia. J Hepatol 2010;52:508–513
Jaroszewicz J, Calle Serrano B, Wursthorn K, et al. Hepatitis B surface antigen (HBsAg) levels in the natural history of hepatitis B virus (HBV)-infection: a European perspective. J Hepatol 2010;52:514–522
Chan HL, Wong VW, Wong GL, et al. A longitudinal study on the natural history of serum hepatitis B surface antigen changes in chronic hepatitis B. Hepatology 2010;52:1232–1241
Tseng TC, Liu CJ, Su TH, et al. Serum hepatitis B surface antigen levels predict surface antigen loss in hepatitis B e antigen seroconverters. Gastroenterology 2011;141:517–525
Chan HL, Wong GL, Tse CH, et al. Viral determinants of hepatitis B surface antigen seroclearance in hepatitis B e antigen-negative chronic hepatitis B patients. J Infect Dis 2011;204:408–414
Tseng TC, Liu CJ, Yang HC, et al. Determinants of spontaneous surface antigen loss in HBeAg-negative patients with a low viral load. Hepatology 2012;55:68–76
Chen YC, Jeng WJ, Chu CM, Liaw YF. Decreasing levels of HBsAg predict HBsAg seroclearance in patients with inactive chronic hepatitis B virus infection. Clin Gastroenterol Hepatol 2012;10:297–302
Livingston SE, Simonetti JP, Bulkow LR, et al. Clearance of hepatitis B e antigen in patients with chronic hepatitis B and genotypes A, B, C, D, and F. Gastroenterology 2007;133:1452–1457
Yu MW, Yeh SH, Chen PJ, et al. Hepatitis B virus genotype and DNA level and hepatocellular carcinoma: a prospective study in men. J Natl Cancer Inst 2005;97:265–272
Thakur V, Guptan RC, Kazim SN, et al. Profile, spectrum and significance of HBV genotypes in chronic liver disease patients in the Indian subcontinent. J Gastroenterol Hepatol 2002;17:165–170
Tanaka Y, Mukaide M, Orito E, et al. Specific mutations in enhancer II/core promoter of hepatitis B virus subgenotypes C1/C2 increase the risk of hepatocellular carcinoma. J Hepatol 2006;45:646–653
Chan HL, Tse CH, Mo F, et al. High viral load and hepatitis B virus subgenotype Ce are associated with increased risk of hepatocellular carcinoma. J Clin Oncol 2008;26:177–182
Zhang HW, Yin JH, Li YT, et al. Risk factors for acute hepatitis B and its progression to chronic hepatitis in Shanghai, China. Gut 2008;57:1713–1720
Chen CH, Hung CH, Lee CM, et al. Pre-S deletion and complex mutations of hepatitis B virus related to advanced liver disease in HBeAg-negative patients. Gastroenterology 2007;133:1466–1474
Yang HI, Yeh SH, Chen PJ, et al. Associations between hepatitis B virus genotype and mutants and the risk of hepatocellular carcinoma. J Natl Cancer Inst 2008;100:1134–1143
Lim SG, Cheng Y, Guindon S, et al. Viral quasi-species evolution during hepatitis Be antigen seroconversion. Gastroenterology 2007;133:951–958
Wang HY, Chien MH, Huang HP, et al. Distinct hepatitis B virus dynamics in the immunotolerant and early immunoclearance phases. J Virol 2010;84:3454–3463
Jung KS, Kim SU, Ahn SH, et al. Risk assessment of hepatitis B virus-related hepatocellular carcinoma development using liver stiffness measurement (FibroScan). Hepatology 2011;53:885–894
Wong GL, Wong VW, Choi PC, et al. Development of a non-invasive algorithm with transient elastography (Fibroscan) and serum test formula for advanced liver fibrosis in chronic hepatitis B. Aliment Pharmacol Ther 2010;31:1095–1103
Fung J, Lai CL, Seto WK, Yuen MF. The use of transient elastography in the management of chronic hepatitis B. Hepatol Int 2011;5:868–875
Yuen MF, Tanaka Y, Fong DY, et al. Independent risk factors and predictive score for the development of hepatocellular carcinoma in chronic hepatitis B. J Hepatol 2009;50:80–88
Wong VW, Chan SL, Mo F, et al. Clinical scoring system to predict hepatocellular carcinoma in chronic hepatitis B carriers. J Clin Oncol 2010;28:1660–1665
Yang HI, Sherman M, Su J, et al. Nomograms for risk of hepatocellular carcinoma in patients with chronic hepatitis B virus infection. J Clin Oncol 2010;28:2437–2444
Yang HI, Yuen MF, Chan HL, et al. Risk estimation for hepatocellular carcinoma in chronic hepatitis B (REACH-B): development and validation of a predictive score. Lancet Oncol 2011;12:568–574
Chien RN. Current therapy for hepatitis C or D or immunodeficiency virus concurrent infection with chronic hepatitis B. Hepatol Int 2008;2:296–303
Piratvisuth T. Immunomodulator therapy of chronic hepatitis B. Hepatol Int 2008;2:140–146
Luo K, Mao Q, Karayiannis P, et al. Tailored regimen of interferon alpha for HBeAg-positive chronic hepatitis B: a prospective controlled study. J Viral Hepat 2008;15:684–689
Lampertico P, Del Ninno E, Vigano M, et al. Long-term suppression of hepatitis B e antigen-negative chronic hepatitis B by 24-month interferon therapy. Hepatology 2003;37:756–763
Papatheodoridis GV, Dimou E, Dimakopoulos K, et al. Outcome of hepatitis B e antigen-negative chronic hepatitis B on long-term nucleos(t)ide analog therapy starting with lamivudine. Hepatology 2005;42:121–129
Cooksley WGE, Piratvisuth T, Lee SD, et al. Peginterferon alpha-2a (40 kDa): an advance in the treatment of hepatitis Be antigen-positive chronic hepatitis B. J Viral Hepatitis 2003;10:298–305
Zhao H, Kurbanov F, Wan MB. Genotype B and younger patient age associated with better response to low-dose therapy: A trial with pegylated/nonpegylated interferon-alpha-2b for hepatitis B e antigen-positive patients with chronic hepatitis B in China. Clin Infect Dis 2007;44:541–548
Lau GK, Piratvisuth T, Luo KX, et al. Peginterferon alfa-2a, lamivudine, and the combination for HBeAg-positive chronic hepatitis B. N Engl J Med 2005;352:2682–2695
Piratvisuth T, Lau GKK, Chao YC, et al. Sustained response to peginterferon alfa-2a (40 kD) with or without lamivudine in Asian patients with HBeAg-positive and HBeAg-negative chronic hepatitis B. Hepatol Int 2008;2:102–110
Chan HL, Leung NW, Hui AY, et al. A randomized, controlled trial of combination therapy for chronic hepatitis B: comparing pegylated interferon-alpha2b and lamivudine with lamivudine alone. Ann Intern Med 2005;15(142):240–250
Liaw YF, Jia JD, Chan HLY, et al. Shorter durations and lower doses of peginterferon alfa-2a are associated with inferior HBeAg seroconversion rates in HBV genotypes B or C. Hepatology 2011;54:1591–1599
Buster EH, Flink HJ, Cakaloglu Y, et al. Sustained HBeAg and HBsAg loss after long-term follow-up of HBeAg-positive patients treated with peginterferon alpha-2b. Gastroenterology 2008;135:459–467
Wong VW, Wong GL, Yan KK, et al. Durability of peginterferon alfa-2b treatment at 5 years in patients with hepatitis B e antigen-positive chronic hepatitis B. Hepatology 2010;51:1945–1953
Marcellin P, Lau GKK, Bonino F, et al. Peginterferon alfa-2 alone, lamivudine alone and the two in combination combination in patients with HBeAg negative chronic hepatitis B. N Engl J Med 2004;351:1206–1217
Papadopoulos VP, Chrysagis DN, Protopapas AN, et al. Peginterferon alfa-2b as monotherapy or in combination with lamivudine in patients with HBeAg-negative chronic hepatitis B: a randomised study. Med Sci Monit 2009;15:CR56–CR61
Marcellin P, Bonino F, Lau GK, et al. Sustained response of hepatitis B e antigen-negative patients 3 years after treatment with peginterferon alpha-2a. Gastroenterology 2009;136:2169–2179
Lampertico P, Viganò M, Colombo M. Treatment of HBeAg-negative chronic hepatitis B with pegylated interferon. Liver Int 2011;31(Suppl 1):90–94
Buster EH, Hansen BE, Buti M, et al. Peginterferon alpha-2b is safe and effective in HBeAg-positive chronic hepatitis B patients with advanced fibrosis. Hepatology 2007;46:388–394
Piccolo P, Lenci I, Demelia L, et al. A randomized controlled trial of pegylated interferon-alpha2a plus adefovir dipivoxil for hepatitis B e antigen negative chronic hepatitis B. Antivir Ther 2009;14:1165–1174
Sarin SK, Sood A, Kumar M, et al. Effect of lowering HBV DNA levels by initial antiviral therapy before adding immunomodulator on treatment of chronic hepatitis B. Am J Gastroenterol 2007;102(1):96–104
Moucari R, Boyer N, Ripault MP, et al. Sequential therapy with adefovir dipivoxil and pegylated interferon alfa-2a for HBeAg-negative patients. J Viral Hepat 2011;18:580–586
Chen CC, Wang PC, Chang HW, Chen CF. Safety and efficacy of two-step peginterferon a-2a treatment in patients of chronic hepatitis B with acute exacerbation. J Viral Hepat 2012;19:161–192
Sonneveld MJ, Wong VW, Woltman AM, et al. Polymorphisms near IL28B and serological response to peginterferon in HBeAg-positive patients with chronic hepatitis B. Gastroenterology 2012;142:513–520
Buster EH, Hansen BE, Lau GK, et al. Factors that predict response of patients with hepatitis B e antigen-positive chronic hepatitis B to peginterferon-alfa. Gastroenterology 2009;137:2002–2009
ter Borg MJ, Hansen BE, Bigot G, et al. ALT and viral load decline during PEG-IFN alpha-2b treatment for HBeAg-positive chronic hepatitis B. J Clin Virol 2008;42:160–164
Liaw YF. On-treatment outcome prediction and adjustment during chronic hepatitis B therapy: now and future. Antivir Ther 2009;14:13–22
Piratvisuth T, Marcellin P, Popescu M, et al. Hepatitis B surface antigen: Association with sustained response to peginterferon alfa-2a in hepatitis B e antigen-positive patients. Hepatol Int 2011; doi:10.1007/s12072-011-9280-0.
Sonneveld MJ, Rijckborst V, Boucher CA, et al. Prediction of sustained response to peginterferon alfa-2b for hepatitis B e antigen-positive chronic hepatitis B using on-treatment hepatitis B surface antigen decline. Hepatology 2010;52:1251–1257
Piratvisuth T, Marcelin P. Further analysis is required to identify an early stopping rule for peginterferon therapy that is valid for all hepatitis B e antigen–positive patients. Hepatology 2011;53:1054–1055
Chien RN, Liaw YF, Chen TC, et al. Efficacy of thymosin alpha-1 in patients with chronic type B hepatitis: a randomized controlled trial. Hepatology 1998;27:1383–1387
Chien RN, Lin CY, Yeh CT, et al. Hepatitis B virus genotype B is associated with better response to thymosin alpha-1 therapy than genotype C. J Viral Hepat 2006;13:845–850
Iino S, Toyota J, Kumada H, et al. The efficacy and safety of thymosin alpha-1 in Japanese patients with chronic hepatitis B; results from a randomized clinical trial. J Viral Hepat 2005;12:300–306
Chan HL, Tang JL, Tam W, et al. The efficacy of thymosin in the treatment of chronic hepatitis B virus infection: a meta-analysis. Aliment Pharmacol Ther 2001;15:1899–1905
Lim SG, Wai CT, Lee YM, et al. A randomized, placebo-controlled trial of thymosin-alpha1 and lymphoblastoid interferon for HBeAg-positive chronic hepatitis B. Antivir Ther 2006;11:245–253
Lee HW, Lee JI, Um SH, et al. Combination therapy of thymosin alpha-1 and lamivudine for HBeAg positive chronic hepatitis B: a prospective randomized, comparative pilot study. J Gastroenterol Hepatol 2008;23:729–735
You J, Zhuang L, Cheng HY, et al. A randomized, controlled, clinical study of thymosin alpha-1 versus interferon-alpha in Chinese patients with chronic hepatitis B lacking hepatitis B envelope antigen. J Chin Med Assoc 2005;68:65–72.
Chang TT, Lai CL, Chien RN, et al. Four years of lamivudine treatment in Chinese patients with chronic hepatitis B. J Gastroenterol Hepatol 2004;19:1276–1282
Yao GB, Zhu M, Cui AY. A 7-year study of lamivudine therapy for chronic hepatitis B virus e antigen-positive chronic hepatitis B patients in China. J Dig Dis 2009;10:131–137
Chien RN, Liaw YF, Atkins M, for Asian Hepatitis Lamivudine Trial Group. Pretherapy alanine transaminase level as a determinant for hepatitis B e antigen seroconversion during lamivudine therapy in patients with chronic hepatitis B. Hepatology 1999;30:770–774
Jonas MM, Little NR, Gardner SD, Members of the International Pediatric Lamivudine Investigator Group. Long-term lamivudine treatment of children with chronic hepatitis B: Durability of therapeutic responses and safety. J Viral Hepat 2008;15:20–27
Lee HW, Lee HJ, Hwang JS, et al. Lamivudine maintenance beyond one year after HBeAg seroconversion is a major factor for sustained virologic response in HBeAg-positive chronic hepatitis B. Hepatology 2010;51:415–421
Kuo YH, Chen CH, Wang JH, et al. Extended lamivudine consolidation therapy in hepatitis B e antigen-positive chronic hepatitis B patients improves sustained hepatitis B e antigen seroconversion. Scand J Gastroenterol 2010;45:75–81
Yeh CT, Hsu CW, Chen YC, Liaw YF. Withdrawal of lamivudine in HBeAg-positive chronic hepatitis B patients after achieving effective maintained virological suppression. J ClinVirol 2009;45:114–118
Chien RN, Yeh CT, Tsai SL, et al. The determinants for sustained HBeAg response to lamivudine therapy. Hepatology 2003;38:1267–1273
Chan HL, Wang H, Niu J, et al. Two-year lamivudine treatment for hepatitis B e antigen-negative chronic hepatitis B: a double-blind, placebo-controlled trial. AntivirTher 2007;12:345–353
Fung SK, Wong F, Hussain M, Lok AS. Sustained response after a 2-year course of lamivudine treatment of hepatitis e antigen-negative chronic hepatitis B. J Viral Hepat 2004;11:432–438
Liu F, Wang L, Li XY, et al. Poor durability of lamivudine effectiveness despite stringent cessation criteria: a prospective clinical study in hepatitis B e antigen-negative chronic hepatitis B patients. J Gastroenterol Hepatol 2011;26:456–460
Chien RN, Liaw YF. Short-term lamivudine therapy in HBeAg-negative chronic active hepatitis B in Taiwan. Antivir Ther 2006;11:947–952
Chan HL, Wong GL, Chim AM, et al. Prediction of off-treatment response to lamivudine by serum hepatitis B surface antigen quantification in hepatitis B e antigen-negative patients. Antivir Ther 2011;16:1249–1257
LiawYF Sung JJ, Chow WC, et al. Lamivudine for patients with chronic hepatitis B and advanced liver disease. N Engl J Med 2004;351:1521–1531
Zoulim F, Locarnini S. Management of treatment failure in chronic hepatitis B. J Hepatol 2012;56 Suppl 1:S112–122
Lai CL, Dienstag J, Schiff E, et al. Prevalence and clinical correlates of YMDD variants during lamivudine therapy for patients with chronic hepatitis B. Clin Infect Dis 2003;36:687–696
Liaw YF, Chien RN, Yeh CT, et al. Acute exacerbation and hepatitis B virus clearance after emergence of YMDD motif mutation during lamivudine therapy. Hepatology 1999;30:567–572
Liaw YF, Gane E, Leung N, et al. 2-year GLOBE trial results: telbivudine is superior to lamivudine in patients with chronic hepatitis B. Gastroenterology 2009;136:486–495
Hou J, Yin YK, Xu D, et al. Telbivudine versus lamivudine in Chinese patients with chronic hepatitis B: results at 1 year of a randomized, double-blind trial. Hepatology 2008;47:447–454
Chan HL, Heathcote EJ, Marcellin P, et al. Treatment of hepatitis B e antigen-positive chronic hepatitis B with telbivudine or adefovir: a randomized trial. Ann Intern Med 2007;147:745–754
Gane E, Wang Y, Liaw YF, et al. Efficacy and safety of prolonged 3-year telbivudine treatment in patients with chronic hepatitis B. Liver Int 2011;31:676–684
Wurthorn K, Jung M, Riva A, et al. Kinetics of hepatitis B surface antigen decline during 3 year of telbivudine treatment in hepatitis B e antigen-positive patients. Hepatology 2010;52:1611–1620
Cai W, Xie Q, An B, et al. On-treatment serum HBsAg level is predictive of sustained off-treatment virologic response to telbivudine in HBeAg-positive chronic hepatitis B patients. J ClinVirol 2010;48:22–26
Zeuzem S, Gane E, Liaw YF, et al. Baseline characteristics and early on-treatment response predict the outcomes of 2 years of telbivudine treatment of chronic hepatitis B. J Hepatol 2009;51:11–20
Marcellin P, Chang TT, Lim SG, et al. Long-term efficacy and safety of adefovir dipivoxil for the treatment of hepatitis B e antigen-positive chronic hepatitis B. Hepatology 2008;48:750–758
Zeng M, Mao Y, Yao GB, et al. Five years of treatment with adefovir dipivoxil in Chinese patients with HBeAg-positive chronic hepatitis B. Liver Int 2012;32:137–146
Hadziyannis SJ, Tassopoulos NC, Heathcote EJ, et al. Long-term therapy with adefovir dipivoxil for HBeAg-negative chronic hepatitis B for up to 5 years. Gastroenterology 2006;131:1743–1751
Liaw YF, Gigi-Raptopoulou M, Cheinquer H, et al. Efficacy and safety of entecavir versus adefovir in chronic hepatitis B patients with hepatic decompensation: a randomized, open-label study. Hepatology 2011;54:91–100
Warner N, Locarnini S. The antiviral drug selected hepatitis B virus rtA181T/sW172* mutant has a dominant negative secretion defect and alters the typical profile of viral rebound. Hepatology 2008;48:88–98
Lee JM, Park JY, Kim DK, et al. Long-term adefovir dipivoxil monotherapy for up to 5 years in lamivudine-resistant chronic hepatitis B. Antivir Ther 2010;15:235–241
Yatsuji H, Suzuki F, Sezaki H, et al. Low risk of adefovir resistance in lamivudine-resistant chronic hepatitis B treated with adefovir plus lamivudine combination therapy: two-year follow-up. J Hepatol 2008;48:923–931
Lampertico P, Vigano M, Manenti E, et al. Adefovir rapidly suppresses hepatitis B in HBeAg-negative patients developing genotypic resistance to lamivudine. Hepatology 2005;42:1414–1419
Liang Y, Jiang J, Su M, et al. Predictors of relapse in chronic hepatitis B after discontinuation of antiviral therapy. Aliment Pharmacol Ther 2011;34:344–352
Marcellin P, Heathcote EJ, Buti M, et al. Tenofovir disoproxilfumarate versus adefovir dipivoxil for chronic hepatitis B. N Engl J Med 2008;359:2442–2455
Heathcote EJ, Marcellin P, Buti M, et al. Three-year efficacy and safety of tenofovir disoproxilfumarate treatment for chronic hepatitis B. Gastroenterology 2011;140:132–143
Liaw YF, Sheen IS, Lee CM, et al. Tenofovir disoproxil fumarate (TDF), emtricitabine/TDF and entecavir in patients with decompensated chronic hepatitis B liver disease. Hepatology 2011;53:62–72
Mauss S, Berger F, Filmann N, et al. Effect of HBV polymerase inhibitors on renal function in patients with chronic hepatitis B. J Hepatol 2011;55:1235–1240
Gutiérrez F, Masiá M. The role of HIV and antiretroviral therapy in bone disease. AIDS Rev 2011;13:109–118
Van Bommel F, de Man R, Wedemeyer H, et al. Long-term efficacy of tenofovir monotherapy for hepatitis B virus-monoinfected patients after failure of nucleoside/nucleotide analogues. Hepatology 2010;51:73–80
Patterson SJ, George J, Strasser SI, et al. Tenofovir disoproxilfumarate rescue therapy following failure of both lamivudine and adefovir dipivoxil in chronic hepatitis B. Gut 2011;60:247–254
Ong A, Wong VW, Wong GL, et al. Management options for lamivudine-resistant chronic hepatitis B patients with suboptimal virological suppression by adefovir. Aliment Pharmacol Ther 2011;34:972–981
Petersen J, Ratziu V, Buti M, et al. Entecavir plus tenofovir combination as rescue therapy in pretreated chronic hepatitis B patients. An international multicenter cohort study. J Hepatol 2012;56:520–526
Chang TT, Lai CL, Yoon SK, et al. Entecavir treatment for up to 5 years in patients with hepatitis B e antigen-positive chronic hepatitis B. Hepatology 2010;51:422–433
Yuen MF, Seto WK, Fung J, et al. Three years of continuous entecavir therapy in treatment-naïve chronic hepatitis B patients: viral suppression, viral resistance and clinical safety. Am J Gastroenterol 2011;106:1264–1271
Zoutendijk R, Peijnders JG, Brown A, et al. Entecavir treatment for chronic hepatitis B: adaptation is not needed for the majority of naïve patients with a partial virological response. Hepatology 2011;54:443–451
Yokosuka O, Takaguchi K, Fujioka S, et al. Long-term use of entecavir in nucleoside-naïve Japanese patients with chronic hepatitis B infection. J Hepatol 2010;52:791–799
Chang TT, Liaw YF, Wu SS, et al. Long-term entecavir therapy results in the reversal of fibrosis/cirrhosis and continued histological improvement in patients with chronic hepatitis B. Hepatology 2010;52:886–893
Fung J, Lai CL, Young J, et al. Quantitative hepatitis B surface antigen levels in patients with chronic hepatitis B after 2 years of entecavir treatment. Am J Gastroenterol 2011;106:1766–1773
Lange CM, Bojunga J, Hofmann WP, et al. Severe lactic acidosis during treatment of chronic hepatitis B with entecavir in patients with impaired liver function. Hepatology 2009;50:2001–2006
Shim JH, Lee JC, Kim KM, et al. Efficacy of entecavir in treatment-naïve patients with hepatitis B virus-related decompensated cirrhosis. J Hepatol 2010;52:176–182
Wong VW, Wong GL, Yiu KK, et al. Entecavir treatment in patients with severe acute exacerbation of chronic hepatitis B. J Hepatol 2011;54:236–242
Tenney DJ, Rose RE, Baldick CJ, et al. Long-term monitoring shows hepatitis B virus resistance to entecavir in nucleoside-naïve patients is rare through 5 years of therapy. Hepatology 2009;49:1503–1514
Flischer RD, Lok AS. Myopathy and neuropathy associated with nucleos(t)ide analog therapy for hepatitis B. J Hepatol 2009;51:787–791
Yuen MF, Han KH, Um SH, et al. Antiviral activity and safety of LB80380 in hepatitis B e antigen-positive chronic hepatitis B patients with lamivudine-resistant disease. Hepatology 2010;51:767–776
Sung JJ, Lai JY, Zeuzem S, et al. Lamivudine compared with lamivudine and adefovir dipivoxil for the treatment of HBeAg-positive chronic hepatitis B. J Hepatol 2008;48:728–735
Si-Ahmed SN, Pradat P, Zoutendijk R, et al. Efficacy and tolerance of a combination of tenofovir disoproxil fumarate plus emtricitabine in patients with chronic hepatitis B: a European multicenter study. Antiviral Res 2011;92:90–95
Hongthanakorn C, Chotiyaputta W, Oberhelman K, et al. Virological breakthrough and resistance in patients with chronic hepatitis B receiving nucleos(t)ide analogues in clinical practice. Hepatology 2011;53:1854–1863
Fontana RJ. Side effects of long-term oral antiviral therapy for hepatitis B. Hepatology 2009;49(5 Suppl):S185–S195
Giles M, Visvanathan K, Sasadeusz J. Antiviral therapy for hepatitis B infection during pregnancy and breastfeeding. Antivir Ther 2011;16:621–628
Shi Z, Yang Y, Ma L, et al. Lamivudine in late pregnancy to interrupt in utero transmission of hepatitis B virus, A systematic review and meta-analysis. Obs Gyn 2010;116:147–159
Han GR, Cao MK, Zhao W, et al. A prospective and open-label study for the efficacy and safety of telbivudine in pregnancy for the prevention of perinatal transmission of hepatitis B virus infection. J Hepatol 2011;55:1215–1221
Petersen J. HBV treatment and pregnancy. J Hepatol 2011;55:1171–1173
Liu CJ, Chuang WL, Lee CM, et al. Peginterferon alfa-2a plus ribavirin for the treatment of dual chronic infection with hepatitis B and C viruses. Gastroenterology 2009;136:496–504
Farci P, Chessa C, Balestrieri C, et al. Treatment of chronic hepatitis D. J Viral Hepat 2007;45:1056–1075
Wedemeyer H, Yurdaydin C, Dalekos GN, et al. Peginterferon plus adefovir versus either drug alone for hepatitis delta. N Engl J Med 2011;364:322–331
Rockstroh JK, Bhagani S, Benhamou Y, et al. European AIDS Clinical Society (EACS) guidelines for the clinical management and treatment of chronic hepatitis B and C coinfection in HIV-infected adults. HIV Med 2008;9:82–88
de Vries-Sluijs TE, Reijnders JG, Hansen BE, et al. Long-term therapy with tenofovir is effective for patients co-infected with human immunodeficiency virus and hepatitis B virus. Gastroenterology 2010;139:1934–1941
Chien RN, Lin CH, Liaw YF. The effect of lamivudine therapy in hepatic decompensation during acute exacerbation of chronic hepatitis B. J Hepatol 2003;38:322–327
Fontana RJ, Hann HW, Perrillo RP, et al. Determinants of early mortality in patients with decompensated chronic hepatitis B treated with antiviral therapy. Gastroenterology 2002;123:719–727
Jonas MM, Block JM, Haber BA, et al. Treatment of children with chronic hepatitis B virus infection in the United States: patient selection and therapeutic options. Hepatology 2010;52:2192–2205
Iorio R, Giannattasio A, Cirillo F, et al. Long-term outcome in children with chronic hepatitis B: a 24-year observation period. Clin Infect Dis 2007;45:943–949
Jeng WJ, Sheen IS, Liaw YF. Hepatitis B virus DNA level predicts hepatic decompensation in patients with acute exacerbation of chronic hepatitis B. Clin Gastroenterol Hepatol 2010;8:541–545
Sun J, Hou JL, Xie Q, et al. Randomised clinical trial: Efficacy of peginterferon alfa-2a in HBeAg positive chronic hepatitis B patients with lamivudine resistance. Aliment Pharmacol Ther 2011;34:424–431
Liu CJ, Chen PJ, Chen DS, Kao JH. Hepatitis B virus reactivation in patients receiving cancer chemotherapy: natural history, pathogenesis, and management. Hepatol Int 2011; doi: 10.1007/s12072-011-9279-6.
Jang JW, Kwon JH, You CR, et al. Risk of HBV reactivation according to viral status and treatment intensity in patients with hepatocellular carcinoma. Antivir Ther 2011;16:969–977
Katz LH, Fraser A, Gafter-Gvili A, et al. Lamivudine prevents reactivation of hepatitis B and reduces mortality in immune suppressed patients: Systematic review and meta-analysis. J Viral Hepat 2008;15:89–102
Li HR, Huang JJ, Guo HQ, et al. Comparison of entecavir and lamivudine in preventing hepatitis B reactivation in lymphoma patients during chemotherapy. J Viral Hepat 2011;18:877–883
Hui CK, Cheung WW, Zhang HY, et al. Kinetics and risk of de novo hepatitis B infection in HBsAg-negative patients undergoing cytotoxic chemotherapy. Gastroenterology 2006;131:59–68
Germanidis G, Hytiroglou P, Zakalka M, Settas L. Reactivation of occult hepatitis B virus infection following treatment of refractory rheumatoid arthritis with abatacept. J Hepatol 2011; doi:10.1016/j.jhep.2011.10.011
Vallet-Pichard A, Fontaine H, Mallet V, Pol S. Viral hepatitis in solid organ transplantation other than liver. J Hepatol 2011;55:474–482
Loomba R, Rowley AK, Wesley R, et al. Hepatitis B immunoglobulin and lamivudine improve hepatitis B-relaated outcomes after liver transplantation: meta-analysis. Clin Gastroenterol Hepatol 2008;6:696–700
Gane EJ, Angus PW, Strasser S, et al. Lamivudine plus low-dose hepatitis B immunoglobulin to prevent recurrent hepatitis B following liver transplantation. Gastroenterology 2007;132:931–937
Angus PW, Patterson SJ, Strasser SI, et al. A randomized study of adefovir dipivoxil in place of HBIG in combination with lamivudine as post-liver transplantation hepatitis B prophylaxis. Hepatology 2008;48:1460–1466
Fung J, Cheung C, Chan SC, et al. Entecavir monotherapy is effective in suppressing hepatitis B virus after liver transplantation. Gastroenterology 2011;141:1212–1219
Patterson SJ, Angus PW. Post-liver transplant hepatitis B prophylaxis: the role of oral nucleos(t)ide analogues. Curr Opin Organ Transplant 2009;14:225–230
Cholongitas E, Papatheodoridis GV, Burroughs AK. Liver grafts from anti-hepatitis B core positive donors: a systematic review. J Hepatol 2010;52:272–279
Wu JC, Huang YH, Chau GY, et al. Risk factors for early and late recurrence in hepatitis B-related hepatocellular carcinoma. J Hepatol 2009;51:890–897
Goto T, Yoshida H, Tateishi R, et al. Influence of serum HBV DNA load on recurrence of hepatocellular carcinoma after treatment with percutaneous radiofrequency ablation. Hepatol Int 2011;5:767–773
Jang JW, Choi JY, Bae SH, et al. A randomized controlled study of preemptive lamivudine in patients receiving transarterial chemo-lipiodolization. Hepatology 2006;43:233–240
Wong JS, Wong GL, Tsoi KK, et al. Meta-analysis: the efficacy of anti-viral therapy in prevention of recurrence after curative treatment of chronic hepatitis B-related hepatocellular carcinoma. Aliment Pharmacol Ther 2011;33:1104–1112
Chan AC, Chok KS, Yuen WK, et al. Impact of antiviral therapy on the survival of patients after major hepatectomy for hepatitis B virus-related hepatocellular carcinoma. Arch Surg 2011;146:675–681
Kim JH, Kwon SY, Lee YS, et al. Virologic response to therapy increases health-related quality of life for patients with chronic hepatitis B. Clin Gastroenterol Hepatol 2012;10:291–296
Chotiyaputta W, Peterson C, Ditah FA, et al. Persistence and adherence to nucleos(t)ide analogue treatment for chronic hepatitis B. J Hepatol 2011;54:12–18
Han KH, Kim DY. Chronic HBV infection with persistently normal ALT: not to treat. Hepatol Int 2008;2:185–189
Liaw YF. Prevention and surveillance of hepatitis B virus-related hepatocellular carcinoma. Semin Liver Dis 2005;25(Suppl 1):40–47
Chu CM, Liaw YF. Chronic hepatitis B virus infection acquired in childhood: special emphasis on prognostic and therapeutic implication of delayed HBeAg seroconversion. J Viral Hepat 2007;14:147–152
Papatheodoridis GV, Manolakopoulos S, Liaw YF, Lok A. Follow-up and indications for liver biopsy in HBeAg-negative chronic hepatitis B virus infection with persistently normal ALT: a systematic review. J Hepatol 2012. doi:10.1016/j.hep.2011.11.030
Hung HF, Chen HH. Cost-effectiveness analysis of prophylactic lamivudine use in preventing vertical transmission of hepatitis B virus infection. Pharmacoeconomics 2011;29:1063–1073
Acknowledgements
The expert pre-meeting was organized and sponsored by the Taiwan Association for the Study of Liver and the Prosperous Foundation, Taipei, Taiwan. The authors thank the secretarial assistance of Ms. Su-Chiung Chu in preparing this statement. The authors also wish to thank Associate Professor Gail Matthews from the Kirby Institute, Sydney, Australia, for her assistance with the sections on HIV-HBV coinfection.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liaw, YF., Kao, JH., Piratvisuth, T. et al. Asian-Pacific consensus statement on the management of chronic hepatitis B: a 2012 update. Hepatol Int 6, 531–561 (2012). https://doi.org/10.1007/s12072-012-9365-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12072-012-9365-4