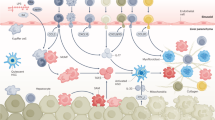
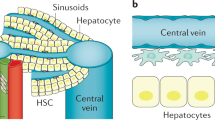
Abstract
Continued elucidation of the mechanisms of hepatic fibrosis has yielded a comprehensive and nuanced portrait of fibrosis progression and regression. The paradigm of hepatic stellate cell (HSC) activation remains the foundation for defining events in hepatic fibrosis and has been complemented by progress in a number of new areas. Cellular sources of extracellular matrix beyond HSCs have been identified. In addition, the role of chemokine, adipokine, neuroendocrine, angiogenic and NAPDH oxidase signaling in the pathogenesis of hepatic fibrosis has been uncovered, as has the contribution of extracellular matrix stiffness to fibrogenesis. There is also increased awareness of the contribution of innate immunity and greater understanding of the complexity of gene regulation in HSCs and myofibroblasts. Finally, both apoptosis and senescence have been recognized as orchestrated programs that eliminate fibrogenic cells during resolution of liver fibrosis. Ironically, the progress that has been made has highlighted the growing disparity between advances in the experimental setting and their translation into new diagnostic tools and treatments. As a result, focus is shifting towards overcoming key translational challenges in order to accelerate the development of new therapies for patients with chronic liver disease.
Key Points
-
Progress in defining the cellular and molecular basis of hepatic fibrosis has brought us to a juncture where translation of these discoveries into diagnostic tools and treatments is nearing reality
-
Novel therapeutic targets continue to be unearthed by new discoveries, including the emergence of the hepatic stellate cell (HSC) as an immunoregulatory cell type
-
HSCs are also now known to participate in adipokine, angiogenic and neuroendocrine signaling, interact with other resident cell types and be regulated by epigenetic and transcriptional mediators
-
Finding better noninvasive markers of fibrogenic activity and disease progression is a high priority in order to accelerate progress in developing antifibrotic drugs
-
Genetic markers that predict fibrosis progression risk, combined with new paradigms to stratify the risk of decompensation among patients with cirrhosis, should improve clinical trial design quality and patient selection for antifibrotic therapies
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Friedman, S. L. Mechanisms of hepatic fibrosis and therapeutic implications. Nat. Clin. Pract. Gastroenterol. Hepatol. 1, 98–105 (2004).
Mitchell, A. E., Colvin, H. M. & Palmer Beasley, R. Institute of Medicine recommendations for the prevention and control of hepatitis B and C. Hepatology 51, 729–733 (2010).
Davis, G. L., Alter, M. J., El-Serag, H., Poynard, T. & Jennings, L. W. Aging of hepatitis C virus (HCV)-infected persons in the United States: a multiple cohort model of HCV prevalence and disease progression. Gastroenterology 138, 513–521, 521 e1–6 (2010).
Lim, Y. S. & Kim, W. R. The global impact of hepatic fibrosis and end-stage liver disease. Clin. Liver Dis. 12, 733–746 (2008).
Cheung, O. & Sanyal, A. J. Recent advances in nonalcoholic fatty liver disease. Curr. Opin. Gastroenterol. 25, 230–237 (2009).
Bugianesi, E. Non-alcoholic steatohepatitis and cancer. Clin. Liver Dis. 11, 191–207 (2007).
Hashizume, H. et al. Primary liver cancers with nonalcoholic steatohepatitis. Eur. J. Gastroenterol. Hepatol. 19, 827–834 (2007).
Jemal, A. et al. Cancer statistics, 2009. CA Cancer J. Clin. 59, 225–249 (2009).
Friedman, S. L. Mechanisms of hepatic fibrogenesis. Gastroenterology 134, 1655–1669 (2008).
Schuppan, D. & Afdhal, N. H. Liver cirrhosis. Lancet 371, 838–851 (2008).
Jiao, J., Friedman, S. L. & Aloman, C. Hepatic fibrosis. Curr. Opin. Gastroenterol. 25, 223–229 (2009).
Parola, M. & Pinzani, M. Hepatic wound repair. Fibrogenesis Tissue Repair 2, 4 (2009).
Henderson, N. C. & Iredale, J. P. Liver fibrosis: cellular mechanisms of progression and resolution. Clin. Sci. (Lond.) 112, 265–280 (2007).
Baertschiger, R. M. et al. Fibrogenic potential of human multipotent mesenchymal stromal cells in injured liver. PLOS ONE 4, e6657 (2009).
Miyata, E. et al. Hematopoietic origin of hepatic stellate cells in the adult liver. Blood 111, 2427–2435 (2008).
Russo, F. P. et al. The bone marrow functionally contributes to liver fibrosis. Gastroenterology 130, 1807–1821 (2006).
Forbes, S. J. et al. A significant proportion of myofibroblasts are of bone marrow origin in human liver fibrosis. Gastroenterology 126, 955–963 (2004).
Fujimiya, T. et al. Pathological roles of bone-marrow-derived stellate cells in a mouse model of alcohol-induced fatty liver. Am. J. Physiol. Gastrointest. Liver Physiol. 297, G974–G980 (2009).
Strieter, R. M. & Mehrad, B. New mechanisms of pulmonary fibrosis. Chest 136, 1364–1370 (2009).
Grande, M. T. & Lopez-Novoa, J. M. Fibroblast activation and myofibroblast generation in obstructive nephropathy. Nat. Rev. Nephrol. 5, 319–328 (2009).
Kisseleva, T. et al. Bone marrow-derived fibrocytes participate in pathogenesis of liver fibrosis. J. Hepatol. 45, 429–438 (2006).
Higashiyama, R. et al. Negligible contribution of bone marrow-derived cells to collagen production during hepatic fibrogenesis in mice. Gastroenterology 137, 1459–1466 (2009).
Kalluri, R. & Neilson, E. G. Epithelial-mesenchymal transition and its implications for fibrosis. J. Clin. Invest. 112, 1776–1784 (2003).
Lin, S. L., Kisseleva, T., Brenner, D. A. & Duffield, J. S. Pericytes and perivascular fibroblasts are the primary source of collagen-producing cells in obstructive fibrosis of the kidney. Am. J. Pathol. 173, 1617–1627 (2008).
Humphreys, B. D. et al. Fate tracing reveals the pericyte and not epithelial origin of myofibroblasts in kidney fibrosis. Am. J. Pathol. 176, 85–97 (2010).
Zeisberg, M. et al. Fibroblasts derive from hepatocytes in liver fibrosis via epithelial to mesenchymal transition. J. Biol. Chem. 282, 23337–23347 (2007).
Omenetti, A. et al. Hedgehog signaling regulates epithelial-mesenchymal transition during biliary fibrosis in rodents and humans. J. Clin. Invest. 118, 3331–3342 (2008).
Omenetti, A. et al. Repair-related activation of hedgehog signaling promotes cholangiocyte chemokine production. Hepatology 50, 518–527 (2009).
Choi, S. S. & Diehl, A. M. Epithelial-to-mesenchymal transitions in the liver. Hepatology 50, 2007–2013 (2009).
Taura, K. et al. Hepatocytes do not undergo epithelial-mesenchymal transition in liver fibrosis in mice. Hepatology 51, 1027–1036 (2010).
Wells, R. G. The epithelial-to-mesenchymal transition in liver fibrosis: here today, gone tomorrow? Hepatology 51, 737–740 (2010).
Kordes, C. et al. CD133+ hepatic stellate cells are progenitor cells. Biochem. Biophys. Res. Commun. 352, 410–417 (2007).
Sawitza, I., Kordes, C., Reister, S. & Haussinger, D. The niche of stellate cells within rat liver. Hepatology 50, 1617–1624 (2009).
Roskams, T. Relationships among stellate cell activation, progenitor cells, and hepatic regeneration. Clin. Liver Dis. 12, 853–860 (2008).
Seki, E. et al. CCR1 and CCR5 promote hepatic fibrosis in mice. J. Clin. Invest. 119, 1858–1870 (2009).
Seki, E. et al. CCR2 promotes hepatic fibrosis in mice. Hepatology 50, 185–197 (2009).
Pinzani, M. & Marra, F. Cytokine receptors and signaling in hepatic stellate cells. Semin. Liver Dis. 21, 397–416 (2001).
Wasmuth, H. E. et al. Antifibrotic effects of CXCL9 and its receptor CXCR3 in livers of mice and humans. Gastroenterology 137, 309–319, 319 e1–3 (2009).
Marra, F. & Bertolani, C. Adipokines in liver diseases. Hepatology 50, 957–969 (2009).
Ikejima, K., Okumura, K., Kon, K., Takei, Y. & Sato, N. Role of adipocytokines in hepatic fibrogenesis. J. Gastroenterol. Hepatol. 22 (Suppl. 1), S87–S92 (2007).
Ding, X. et al. The roles of leptin and adiponectin: a novel paradigm in adipocytokine regulation of liver fibrosis and stellate cell biology. Am. J. Pathol. 166, 1655–1669 (2005).
Ebrahimkhani, M. R., Elsharkawy, A. M. & Mann, D. A. Wound healing and local neuroendocrine regulation in the injured liver. Expert Rev. Mol. Med. 10, e11 (2008).
Teixeira-Clerc, F. et al. CB1 cannabinoid receptor antagonism: a new strategy for the treatment of liver fibrosis. Nat. Med. 12, 671–676 (2006).
Munoz-Luque, J. et al. Regression of fibrosis after chronic stimulation of cannabinoid CB2 receptor in cirrhotic rats. J. Pharmacol. Exp. Ther. 324, 475–483 (2008).
Deveaux, V. et al. Cannabinoid CB2 receptor potentiates obesity-associated inflammation, insulin resistance and hepatic steatosis. PLoS ONE 4, e5844 (2009).
Passino, M. A., Adams, R. A., Sikorski, S. L. & Akassoglou, K. Regulation of hepatic stellate cell differentiation by the neurotrophin receptor p75NTR. Science 315, 1853–1856 (2007).
Kendall, T. J. et al. p75 Neurotrophin receptor signaling regulates hepatic myofibroblast proliferation and apoptosis in recovery from rodent liver fibrosis. Hepatology 49, 901–910 (2009).
Fernandez, M. et al. Angiogenesis in liver disease. J. Hepatol. 50, 604–620 (2009).
Novo, E. & Parola, M. Redox mechanisms in hepatic chronic wound healing and fibrogenesis. Fibrogenesis Tissue Repair 1, 5 (2008).
De Minicis, S. & Brenner, D. A. NOX in liver fibrosis. Arch. Biochem. Biophys. 462, 266–272 (2007).
Zhan, S. S. et al. Phagocytosis of apoptotic bodies by hepatic stellate cells induces NADPH oxidase and is associated with liver fibrosis in vivo. Hepatology 43, 435–443 (2006).
Colmenero, J. et al. Effects of losartan on hepatic expression of non-phagocytic NADPH oxidase and fibrogenic genes in patients with chronic hepatitis C. Am. J. Physiol. Gastrointest. Liver Physiol. 297, G726–G734 (2009).
Moreno, M. et al. Reduction of advanced liver fibrosis by short-term targeted delivery of an angiotensin receptor blocker to hepatic stellate cells in rats. Hepatology 51, 942–952 (2009).
Sanyal, A. J. et al. Pioglitazone, vitamin E, or placebo for nonalcoholic steatohepatitis. N. Engl. J. Med. 362, 1–5 (2010).
Sanyal, A. A randomized controlled trial of pioglitazone or vitamin E for nonalcoholic steatohepatitis (PIVENS) [abstract]. Hepatology 50, 90A (2009).
Wells, R. G. The role of matrix stiffness in regulating cell behavior. Hepatology 47, 1394–1400 (2008).
Georges, P. C. et al. Increased stiffness of the rat liver precedes matrix deposition: implications for fibrosis. Am. J. Physiol. Gastrointest. Liver Physiol. 293, G1147–G1154 (2007).
Park, O. et al. Diverse roles of invariant natural killer T cells in liver injury and fibrosis induced by carbon tetrachloride. Hepatology 49, 1683–1694 (2009).
Gao, B., Radaeva, S. & Park, O. Liver natural killer and natural killer T cells: immunobiology and emerging roles in liver diseases. J. Leukoc. Biol. 86, 513–528 (2009).
Notas, G., Kisseleva, T. & Brenner, D. NK and NKT cells in liver injury and fibrosis. Clin. Immunol. 130, 16–26 (2009).
Connolly, M. K. et al. In liver fibrosis, dendritic cells govern hepatic inflammation in mice via TNF-alpha. J. Clin. Invest. 119, 3213–3225 (2009).
Seki, E. et al. TLR4 enhances TGF-beta signaling and hepatic fibrosis. Nat. Med. 13, 1324–1332 (2007).
Seki, E. & Brenner, D. A. Toll-like receptors and adaptor molecules in liver disease: update. Hepatology 48, 322–335 (2008).
Guo, J. et al. Functional linkage of cirrhosis-predictive single nucleotide polymorphisms of Toll-like receptor 4 to hepatic stellate cell responses. Hepatology 49, 960–968 (2009).
Mencin, A., Kluwe, J. & Schwabe, R. F. Toll-like receptors as targets in chronic liver diseases. Gut 58, 704–720 (2009).
Chakraborty, J. B. & Mann, D. A. NF-kappaB signalling: embracing complexity to achieve translation. J. Hepatol. 52, 285–291 (2010).
Watanabe, A. et al. Inflammasome-mediated regulation of hepatic stellate cells. Am. J. Physiol. Gastrointest. Liver Physiol. 296, G1248–G1257 (2009).
Muhanna, N. et al. Activation of hepatic stellate cells after phagocytosis of lymphocytes: a novel pathway of fibrogenesis. Hepatology 48, 963–977 (2008).
Holt, A. P. et al. Liver myofibroblasts regulate infiltration and positioning of lymphocytes in human liver. Gastroenterology 136, 705–714 (2009).
Mann, J. & Mann, D. A. Transcriptional regulation of hepatic stellate cells. Adv. Drug Deliv. Rev. 61, 497–512 (2009).
Mann, J. et al. Regulation of myofibroblast transdifferentiation by DNA methylation and MeCP2: implications for wound healing and fibrogenesis. Cell Death Differ. 14, 275–285 (2007).
Mann, J. et al. MeCP2 controls an epigenetic pathway that promotes myofibroblast transdifferentiation and fibrosis. Gastroenterology 138, 705–714, 714 e1–4 (2010).
Fritz, D. & Stefanovic, B. RNA-binding protein RBMS3 is expressed in activated hepatic stellate cells and liver fibrosis and increases expression of transcription factor Prx1. J. Mol. Biol. 371, 585–595 (2007).
Guo, C. J., Pan, Q., Li, D. G., Sun, H. & Liu, B. W. miR-15b and miR-16 are implicated in activation of the rat hepatic stellate cell: an essential role for apoptosis. J. Hepatol. 50, 766–778 (2009).
Venugopal, S. K. et al. Liver fibrosis causes down-regulation of miRNA-150 and miRNA-194 in hepatic stellate cells and their over-expression causes decreased stellate cell activation. Am. J. Physiol. Gastrointest. Liver Physiol. 298, G101–G106 (2009).
Oakley, F. et al. Angiotensin II activates I kappaB kinase phosphorylation of RelA at Ser 536 to promote myofibroblast survival and liver fibrosis. Gastroenterology 136, 2334–2344 e1 (2009).
Buck, M. & Chojkier, M. A ribosomal S-6 kinase-mediated signal to C/EBP-beta is critical for the development of liver fibrosis. PLoS ONE 2, e1372 (2007).
Friedman, S. L. & Bansal, M. B. Reversal of hepatic fibrosis—fact or fantasy? Hepatology 43, S82–S88 (2006).
Mallet, V. et al. Brief communication: the relationship of regression of cirrhosis to outcome in chronic hepatitis C. Ann. Intern. Med. 149, 399–403 (2008).
Gonzalez, S. A. et al. Inverse association between hepatic stellate cell apoptosis and fibrosis in chronic hepatitis C virus infection. J. Viral Hepat. 16, 141–148 (2009).
Oakley, F. et al. Inhibition of inhibitor of kappaB kinases stimulates hepatic stellate cell apoptosis and accelerated recovery from rat liver fibrosis. Gastroenterology 128, 108–120 (2005).
Jiang, J. X., Mikami, K., Venugopal, S., Li, Y. & Torok, N. J. Apoptotic body engulfment by hepatic stellate cells promotes their survival by the JAK/STAT and Akt/NF-kappaB-dependent pathways. J. Hepatol. 51, 139–148 (2009).
Krizhanovsky, V. et al. Senescence of activated stellate cells limits liver fibrosis. Cell 134, 657–667 (2008).
Regev, A. et al. Sampling error and intraobserver variation in liver biopsy in patients with chronic HCV infection. Am. J. Gastroenterol. 97, 2614–2618 (2002).
Ratziu, V. et al. Sampling variability of liver biopsy in nonalcoholic fatty liver disease. Gastroenterology 128, 1898–1906 (2005).
Bedossa, P., Dargere, D. & Paradis, V. Sampling variability of liver fibrosis in chronic hepatitis C. Hepatology 38, 1449–1457 (2003).
Goodman, Z. D. et al. Fibrosis progression in chronic hepatitis C: morphometric image analysis in the HALT-C trial. Hepatology 50, 1738–1749 (2009).
Calvaruso, V. et al. Computer-assisted image analysis of liver collagen: relationship to Ishak scoring and hepatic venous pressure gradient. Hepatology 49, 1236–1244 (2009).
Everhart, J. E. et al. Prognostic value of Ishak fibrosis stage: findings from the hepatitis C antiviral long-term treatment against cirrhosis trial. Hepatology 51, 585–594 (2010).
Castera, L. Transient elastography and other noninvasive tests to assess hepatic fibrosis in patients with viral hepatitis. J. Viral Hepat. 16, 300–314 (2009).
Vizzutti, F., Arena, U., Marra, F. & Pinzani, M. Elastography for the non-invasive assessment of liver disease: limitations and future developments. Gut 58, 157–160 (2009).
Pinzani, M., Vizzutti, F., Arena, U. & Marra, F. Technology Insight: noninvasive assessment of liver fibrosis by biochemical scores and elastography. Nat. Clin. Pract. Gastroenterol. Hepatol. 5, 95–106 (2008).
Arena, U. et al. Acute viral hepatitis increases liver stiffness values measured by transient elastography. Hepatology 47, 380–384 (2008).
Vigano, M. et al. Transient elastography assessment of the liver stiffness dynamics during acute hepatitis B. Eur. J. Gastroenterol. Hepatol. 22, 180–184 (2009).
Carrion, J. A. et al. Liver stiffness identifies two different patterns of fibrosis progression in patients with hepatitis C virus recurrence after liver transplantation. Hepatology 51, 23–34 (2010).
Talwalkar, J. A. et al. Magnetic resonance imaging of hepatic fibrosis: emerging clinical applications. Hepatology 47, 332–342 (2008).
Bonekamp, S., Kamel, I., Solga, S. & Clark, J. Can imaging modalities diagnose and stage hepatic fibrosis and cirrhosis accurately? J. Hepatol. 50, 17–35 (2009).
Taouli, B. et al. Diffusion-weighted MRI for quantification of liver fibrosis: preliminary experience. AJR Am. J. Roentgenol. 189, 799–806 (2007).
Hagiwara, M. et al. Advanced liver fibrosis: diagnosis with 3D whole-liver perfusion MR imaging--initial experience. Radiology 246, 926–934 (2008).
Barash, H. et al. Functional magnetic resonance imaging monitoring of pathological changes in rodent livers during hyperoxia and hypercapnia. Hepatology 48, 1232–1241 (2008).
Fahey, B. J., Nightingale, K. R., Nelson, R. C., Palmeri, M. L. & Trahey, G. E. Acoustic radiation force impulse imaging of the abdomen: demonstration of feasibility and utility. Ultrasound Med. Biol. 31, 1185–1198 (2005).
Friedrich-Rust, M. et al. Liver fibrosis in viral hepatitis: noninvasive assessment with acoustic radiation force impulse imaging versus transient elastography. Radiology 252, 595–604 (2009).
Smith, J. O. & Sterling, R. K. Systematic review: Non-invasive methods of fibrosis analysis in chronic hepatitis C. Aliment. Pharmacol. Ther. 30, 557–576 (2009).
Smith, M. W. et al. Gene expression patterns that correlate with hepatitis C and early progression to fibrosis in liver transplant recipients. Gastroenterology 130, 179–187 (2006).
Leroy, V. et al. Prospective comparison of six non-invasive scores for the diagnosis of liver fibrosis in chronic hepatitis C. J. Hepatol. 46, 775–782 (2007).
Gressner, O. A., Weiskirchen, R. & Gressner, A. M. Biomarkers of hepatic fibrosis, fibrogenesis and genetic pre-disposition pending between fiction and reality. J. Cell. Mol. Med. 11, 1031–1051 (2007).
Nunes, D. et al. Noninvasive markers of liver fibrosis are highly predictive of liver-related death in a cohort of HCV-infected individuals with and without HIV infection. Am. J. Gastroenterol. doi: 10.1038/ajg.2009.746.
Ngo, Y. et al. A prospective analysis of the prognostic value of biomarkers (FibroTest) in patients with chronic hepatitis C. Clin. Chem. 52, 1887–1896 (2006).
Mayo, M. J. et al. Prediction of clinical outcomes in primary biliary cirrhosis by serum enhanced liver fibrosis assay. Hepatology 48, 1549–1557 (2008).
Sebastiani, G. et al. SAFE biopsy: a validated method for large-scale staging of liver fibrosis in chronic hepatitis C. Hepatology 49, 1821–1827 (2009).
Castera, L. et al. Prospective comparison of transient elastography, Fibrotest, APRI, and liver biopsy for the assessment of fibrosis in chronic hepatitis C. Gastroenterology 128, 343–350 (2005).
Fontana, R. J. et al. Serum fibrosis marker levels decrease after successful antiviral treatment in chronic hepatitis C patients with advanced fibrosis. Clin. Gastroenterol. Hepatol. 7, 219–226 (2009).
Nobili, V. et al. Performance of ELF serum markers in predicting fibrosis stage in pediatric non-alcoholic fatty liver disease. Gastroenterology 136, 160–167 (2009).
de Ledinghen, V. et al. Diagnosis of hepatic fibrosis and cirrhosis by transient elastography in HIV/hepatitis C virus-coinfected patients. J. Acquir. Immune Defic. Syndr. 41, 175–179 (2006).
Sanchez-Conde, M. et al. Comparison of transient elastography and liver biopsy for the assessment of liver fibrosis in HIV/hepatitis C virus-coinfected patients and correlation with noninvasive serum markers. J. Viral Hepat. 17, 280–286 (2009).
Berenguer, J. et al. Sustained virological response to interferon plus ribavirin reduces liver-related complications and mortality in patients coinfected with human immunodeficiency virus and hepatitis C virus. Hepatology 50, 407–413 (2009).
Russo, M. W. et al. Early hepatic stellate cell activation is associated with advanced fibrosis after liver transplantation in recipients with hepatitis C. Liver Transpl. 11, 1235–1241 (2005).
Armuzzi, A. et al. Review article: breath testing for human liver function assessment. Aliment. Pharmacol. Ther. 16, 1977–1996 (2002).
Petrolati, A. et al. 13C-methacetin breath test for monitoring hepatic function in cirrhotic patients before and after liver transplantation. Aliment. Pharmacol. Ther. 18, 785–790 (2003).
Ripoll, C. et al. Hepatic venous pressure gradient predicts clinical decompensation in patients with compensated cirrhosis. Gastroenterology 133, 481–488 (2007).
Talwalkar, J. A. Antifibrotic therapies—emerging biomarkers as treatment end points. Nat. Rev. Gastroenterol. Hepatol. 7, 59–61 (2010).
Hold, G. L., Untiveros, P., Saunders, K. A. & El-Omar, E. M. Role of host genetics in fibrosis. Fibrogenesis Tissue Repair 2, 6 (2009).
Weber, S., Gressner, O. A., Hall, R., Grunhage, F. & Lammert, F. Genetic determinants in hepatic fibrosis: from experimental models to fibrogenic gene signatures in humans. Clin. Liver Dis. 12, 747–757 (2008).
Wasmuth, H. E. et al. The Marburg I variant (G534E) of the factor VII-activating protease determines liver fibrosis in hepatitis C infection by reduced proteolysis of platelet-derived growth factor BB. Hepatology 49, 775–780 (2009).
Asselah, T. et al. Genetics, genomics, and proteomics: implications for the diagnosis and the treatment of chronic hepatitis C. Semin. Liver Dis. 27, 13–27 (2007).
Huang, H. et al. A 7 gene signature identifies the risk of developing cirrhosis in patients with chronic hepatitis C. Hepatology 46, 297–306 (2007).
Huang, H. et al. Identification of two gene variants associated with risk of advanced fibrosis in patients with chronic hepatitis C. Gastroenterology 130, 1679–1687 (2006).
Marcolongo, M. et al. A seven-gene signature (cirrhosis risk score) predicts liver fibrosis progression in patients with initially mild chronic hepatitis C. Hepatology 50, 1038–1044 (2009).
Wasmuth, H. E. et al. CC chemokine receptor 5 delta32 polymorphism in two independent cohorts of hepatitis C virus infected patients without hemophilia. J. Mol. Med. 82, 64–69 (2004).
Hillebrandt, S. et al. Complement factor 5 is a quantitative trait gene that modifies liver fibrogenesis in mice and humans. Nat. Genet. 37, 835–843 (2005).
Hung, T. M. et al. A novel nonsynonymous variant of matrix metalloproteinase-7 confers risk of liver cirrhosis. Hepatology 50, 1184–1193 (2009).
Freedman, N. D. et al. Coffee intake is associated with lower rates of liver disease progression in chronic hepatitis C. Hepatology 50, 1360–1369 (2009).
Ruhl, C. E. & Everhart, J. E. Coffee and tea consumption are associated with a lower incidence of chronic liver disease in the United States. Gastroenterology 129, 1928–1936 (2005).
Modi, A. A. et al. Increased caffeine consumption is associated with reduced hepatic fibrosis. Hepatology 51, 201–209 (2010).
Chan, E. S. et al. Adenosine A(2A) receptors play a role in the pathogenesis of hepatic cirrhosis. Br. J. Pharmacol. 148, 1144–1155 (2006).
Chan, H. L. et al. Hepatitis B virus genotype C is associated with more severe liver fibrosis than genotype B. Clin. Gastroenterol. Hepatol. 7, 1361–1366 (2009).
Bochud, P. Y. et al. Genotype 3 is associated with accelerated fibrosis progression in chronic hepatitis C. J. Hepatol. 51, 655–666 (2009).
Macias, J. et al. Fast fibrosis progression between repeated liver biopsies in patients coinfected with human immunodeficiency virus/hepatitis C virus. Hepatology 50, 1056–1063 (2009).
Pinzani, M. & Vizzutti, F. Fibrosis and cirrhosis reversibility: clinical features and implications. Clin. Liver Dis. 12, 901–913 (2008).
Ramachandran, P. & Iredale, J. P. Reversibility of liver fibrosis. Ann. Hepatol. 8, 283–291 (2009).
Garcia-Tsao, G., Friedman, S., Iredale, J. & Pinzani, M. Now there are many (stages) where before there was one: In search of a pathophysiological classification of cirrhosis. Hepatology 51, 1445–1449 (2010).
Ghany, M. G. et al. Predicting clinical and histologic outcomes based on standard laboratory tests in advanced chronic hepatitis C. Gastroenterology 138, 136–146 (2010).
Kazemi, F. et al. Liver stiffness measurement selects patients with cirrhosis at risk of bearing large oesophageal varices. J. Hepatol. 45, 230–235 (2006).
Masuzaki, R. et al. Risk assessment of hepatocellular carcinoma in chronic hepatitis C patients by transient elastography. J. Clin. Gastroenterol. 42, 839–843 (2008).
Nagula, S., Jain, D., Groszmann, R. J. & Garcia-Tsao, G. Histological–hemodynamic correlation in cirrhosis—a histological classification of the severity of cirrhosis. J. Hepatol. 44, 111–117 (2006).
Iredale, J. P. Models of liver fibrosis: exploring the dynamic nature of inflammation and repair in a solid organ. J. Clin. Invest. 117, 539–548 (2007).
Popov, Y., Patsenker, E., Fickert, P., Trauner, M. & Schuppan, D. Mdr2 (Abcb4)−/− mice spontaneously develop severe biliary fibrosis via massive dysregulation of pro- and antifibrogenic genes. J. Hepatol. 43, 1045–1054 (2005).
Lichtman, S. N., Wang, J. & Clark, R. L. A microcholangiographic study of liver disease models in rats. Acad. Radiol. 2, 515–521 (1995).
Popov, Y. et al. Integrin alphavbeta6 is a marker of the progression of biliary and portal liver fibrosis and a novel target for antifibrotic therapies. J. Hepatol. 48, 453–464 (2008).
Friedman, S. L. Reversibility of hepatic fibrosis and cirrhosis--is it all hype? Nat. Clin. Pract. Gastroenterol. Hepatol. 4, 236–237 (2007).
Ghiassi-Nejad, Z. & Friedman, S. L. Advances in antifibrotic therapy. Expert Rev. Gastroenterol. Hepatol. 2, 803–816 (2008).
Rockey, D. C. Current and future anti-fibrotic therapies for chronic liver disease. Clin. Liver Dis. 12, 939–962 (2008).
Popov, Y. & Schuppan, D. Targeting liver fibrosis: strategies for development and validation of antifibrotic therapies. Hepatology 50, 1294–1306 (2009).
Schoemaker, M. H. et al. PDGF-receptor beta-targeted adenovirus redirects gene transfer from hepatocytes to activated stellate cells. Mol. Pharm. 5, 399–406 (2008).
Gonzalo, T. et al. Local inhibition of liver fibrosis by specific delivery of a PDGF kinase inhibitor to hepatic stellate cells. J. Pharmacol. Exp. Ther. 321, 856–865 (2007).
Friedman, S. L. Targeting siRNA to arrest fibrosis. Nat. Biotechnol. 26, 399–400 (2008).
Sato, Y. et al. Resolution of liver cirrhosis using vitamin A-coupled liposomes to deliver siRNA against a collagen-specific chaperone. Nat. Biotechnol. 26, 431–442 (2008).
Acknowledgements
Work in the author's laboratory is supported by NIH grants RO1DK37340, RO1DK56621, P20AA017067 and 1K05AA018408-01. This article highlights primarily recent studies and review articles of general interest to the readership. As a result many outstanding publications in the field could not be cited because of space constraints.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
Scott L. Friedman is a consultant for Abbott, Amgen, Axcan Pharma, Human Genome Sciences, MDRNA, Onyx Pharmaceuticals, Salix Pharmaceuticals, Sanofi Aventis and Siemens Diagnostics (each less than US$5,000 per annum). He has stock ownership/equity and is a consultant for Angion Biomedica, Conatus Pharmaceuticals, Exalenz and Intercept Pharmaceuticals (each less than US$10,000 and less than 5%). He has restricted grants and contracts with Celera (to study the mechanisms linking fibrosis risk SNPs to pathogenesis) and Bayer/Onyx Pharmaceuticals (to study antifibrotic activity of sorafenib in hepatic fibrosis). He also holds US patents on noninvasive agents for diagnosis and prognosis of the progression of fibrosis (US Patent no. 6,264,949 B1) and KLF6, a novel growth inhibitory and tumor suppressor gene (US Patent no. US2005/0181374), and has four other US patents pending that relate to hepatic fibrosis and/or hepatocellular carcinoma.
Rights and permissions
About this article
Cite this article
Friedman, S. Evolving challenges in hepatic fibrosis. Nat Rev Gastroenterol Hepatol 7, 425–436 (2010). https://doi.org/10.1038/nrgastro.2010.97
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrgastro.2010.97
This article is cited by
-
LncRNA H19-EZH2 interaction promotes liver fibrosis via reprogramming H3K27me3 profiles
Acta Pharmacologica Sinica (2023)
-
Ligand-tethered lipid nanoparticles for targeted RNA delivery to treat liver fibrosis
Nature Communications (2023)
-
The chromatin remodeling protein BRG1 regulates HSC-myofibroblast differentiation and liver fibrosis
Cell Death & Disease (2023)
-
Downregulation of miR-20b-5p Contributes to the Progression of Liver Fibrosis via the STAT3 Signaling Pathway In Vivo and In Vitro
Digestive Diseases and Sciences (2023)
-
Identification of a rare Gli1+ progenitor cell population contributing to liver regeneration during chronic injury
Cell Discovery (2022)