Abstract
Vitamin D analogues with reduced hypercalcemic activity are under clinical investigation for use against colon cancer and other neoplasias. However, only a subset of patients responds to this therapy, most probably due to loss of vitamin D receptor (VDR) expression during tumour progression. Recent data show that SNAIL transcription factor represses VDR expression, and thus abolishes the antiproliferative and prodifferentiation effects of VDR ligands in cultured cancer cells and their antitumour action in xenografted mice. Accordingly, upregulation of SNAIL in human colon tumours associates with downregulation of VDR. These findings suggest that SNAIL may be associated with loss of responsiveness to vitamin D analogues and may thus be used as an indicator of patients who are unlikely to respond to this therapy.
Similar content being viewed by others
Main
Colorectal cancer is the most frequent neoplasia and the second leading cause of cancer death in Europe, with 218 000 new cases diagnosed each year and an overall rate of 58.1 cases per 100 000 person-years (Ferlay et al, 1999). By sex, it constitutes the second most frequent tumour in women after breast cancer and the third in men after lung and prostate tumours. Surgical resection is the therapy of choice for localised tumours, which is curative at an early stage of the disease. However, there is no satisfactory treatment for advanced colon cancer despite the improvement in systemic therapy and radiotherapy in the last decades.
Vitamin D intake and sunlight exposure show a protective effect against colorectal carcinogenesis (Peters et al, 2001, and refs. therein). A recent study suggests that vitamin D and calcium supplementation act together to reduce colorectal adenoma risk (Grau et al, 2003). In line with epidemiological data, 1α,25-dihydroxyvitamin D3 (1,25(OH)2D3, Calcitriol), the most active metabolite of vitamin D, has antiproliferative, proapoptotic and prodifferentiation effects on many tumour cells in vitro (Figure 1), including cells of the large intestine, in addition to its classical role regulating calcium and phosphate homeostasis and bone biology (Hansen et al, 2001; Lamprecht and Lipkin, 2003). Remarkably, some vitamin D analogues enhance the cytotoxic effects of chemotherapeutic drugs and radiotherapy.
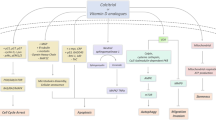
Effects of 1,25(OH)2D3 in colon cancer cells. SNAIL inhibits 1,25(OH)2D3 action by repressing VDR gene expression. Discontinuous lanes refer to not confirmed processes. 1,25(OH)2D3 inhibits β-catenin signalling (Pálmer et al, 2001) and up- or down-regulates numerous genes, directly or indirectly (Lamprecht and Lipkin, 2003; Pálmer et al, 2003).
The clinical use of 1,25(OH)2D3 is hampered by its hypercalcemic effect. For this reason, a number of 1,25(OH)2D3 analogues (deltanoids) with variable biological activity and reduced calcemic properties have been synthesised (Carlberg and Mouriño, 2003). Several of them show promising anticancer action in preclinical studies and are currently in clinical trials (Hansen et al, 2001). The use of vitamin D analogues for prevention or therapy in cancer patients is still in its infancy. Few Phase I and II studies have been reported, in general showing acceptable toxicity but activity only in a subset of patients.
1,25(OH)2D3 synthesis and action
Dietary vitamin D is scarce, and most vitamin D3 is synthesised in the skin by the action of sunlight. Ultraviolet-B radiation converts 7-dehydrocholesterol to previtamin D3, which thermoisomerises to vitamin D3. This is subjected to two consecutive hydroxylation reactions: first in the liver by vitamin D3 25-hydroxylase (CYP27A1) and then in the kidney by 25-hydroxyvitamin D3-1α-hydroxylase (CYP27B1) to render 1,25(OH)2D3. Recently, 25-hydroxyvitamin D3-1α-hydroxylase has also been found to be expressed by osteoblasts, keratinocytes, chondrocytes, macrophages and several epithelia including the intestinal (MacDonald et al, 2001). 1,25(OH)2D3 is converted to the much less active 1,24,25(OH)3D3 compound by the 24-hydroxylase (CYP24) (MacDonald et al, 2001).
Vitamin D receptor (VDR) is a ligand-regulated transcription factor that mediates most, if not all, the biological effects of 1,25(OH)2D3 (MacDonald et al, 2001). Most 1,25(OH)2D3 analogues act also via VDR, although some have effects unrelated to VDR binding such as CYP24 enzyme inhibition (Posner et al, 2004). For years considered to be exclusively nuclear, VDR is now believed to shuttle constantly between nucleus and cytoplasm; ligand binding and its interaction with the retinoid X receptor (RXR) increase nuclear accumulation (Prüfer and Barsony, 2002). Unliganded VDR binds transcriptional corepressors such as SMRT, N-CoR or Alien, actively repressing target genes. Hormone binding induces a conformational change in VDR that increases the formation of VDR-RXR heterodimers and DNA binding, and causes corepressor release. Subsequently, VDR binds coactivators (SRC-1, ACTR, GRIP-1 and SKIP, among others) and interacts with the multiprotein DRIP (vitamin-D-receptor-interacting protein) and chromatin remodeller complexes, which finally leads to activation of genes mediating most hormone actions (MacDonald et al, 2001). Furthermore, VDR is phosphorylated at several residues by different kinases, which contribute to the regulation of its localisation and function in ways that remain to be understood.
In addition to the genomic pathway involving nuclear VDR, a nongenomic pathway of 1,25(OH)2D3 action has been postulated (Figure 1). This latter pathway is characterised by rapid, gene expression-independent effects such as changes in ion levels or the activity of some kinases, which might be mediated by membrane or cytosolic VDR or by other unknown receptors (Lamprecht and Lipkin, 2003, and references therein).
1,25(OH)2D3 and colon cancer
The predominant effect of 1,25(OH)2D3, be it antiproliferative, proapoptotic or prodifferentiation, depends on the cell type, and is supposedly based on the set of genes regulated in each particular system. Cell-cycle arrest may result from the induction of cyclin-dependent kinase inhibitors such as p21CIP and p27KIP and the repression of cyclin D1, whereas the activation of BAX and BAK and the inhibition of BCL2 and IAP contribute to apoptotic induction (Figure 1) (Lamprecht and Lipkin, 2003).
Mutation of APC (adenomatous polyposis coli) tumour suppressor gene or, less frequently, of CTNNB1 encoding β-catenin, a component of adherens junctions involved in intercellular adhesion, is a common early alteration in human colon cancer (Giles et al, 2003). These mutations, or stimulation of the signaling pathway by Wnt extracellular factors, prevent β-catenin protein degradation by a complex involving APC, Axin and the protein kinases GSK-3β and CKIα, causing its accumulation in the cytosol and translocation into the nucleus (Giles et al, 2003). In the nucleus, β-catenin forms complexes with the TCF/LEF family of transcriptional regulators leading to the activation of genes involved in proliferation and invasiveness, thus promoting colon cancer progression (Giles et al, 2003 and references therein).
A study by our group has revealed that 1,25(OH)2D3 and several nonhypercalcemic analogues promote the differentiation and inhibit the proliferation of human colon cancer cells expressing high VDR levels (SW480-ADH) through the induction of E-cadherin and other adhesion proteins and the blockade of β-catenin transcriptional activity (Pálmer et al, 2001) (Figure 1). As a result, 1,25(OH)2D3 inhibits the expression of β-catenin target genes such as c-MYC, TCF1 or CD44 (Pálmer et al, 2001). In total, 200 1,25(OH)2D3 target genes have been reported in SW480-ADH cells: they are involved in different aspects of cell biology and support the beneficial effect of the hormone on cell phenotype and growth (Pálmer et al, 2003). In summary, 1,25(OH)2D3 regulates many genes either directly, through VDR binding to their regulatory regions, or indirectly, via intermediate genes or by affecting other pathways, such as Wnt/β-catenin, that are related to its antitumoral action.
VDR and colon cancer
In the human colon, normal epithelial cells and some cancer cells express VDR (Kállay et al, 2002). Furthermore, some colon cancer cells express 25-hydroxyvitamin D3-1α-hydroxylase and can thus synthesise 1,25(OH)2D3 from its precursor (Cross et al, 2001). This suggests that the VDR expression observed in low-grade tumours and the tumour-localised production of this hormone could be an autocrine/paracrine means to control tumour progression. Concordantly, a high level of VDR expression is associated with a favourable prognosis in colorectal cancer (Cross et al, 1996; Evans et al, 1998). Vitamin D receptor expression is enhanced during early stages of colon cancer but, analogously to oestrogen and progesterone receptors in breast cancer, it is downregulated during late colon cancer progression (Sheinin et al, 2000; Cross et al, 2001), causing ligand unresponsiveness and, possibly, failure of therapy with vitamin D analogues (Figure 2). Some studies indicate that VDR expression decreases in high-grade carcinomas to levels found in normal mucosa (Sheinin et al, 2000; Cross et al, 2001; Kállay et al, 2002), while others found diminished VDR expression already in low- and intermediate-grade tumours and a decrease below normal mucosa levels in high-grade carcinomas (Pálmer et al, 2004) (Figure 2).
Opposite pattern of expression of VDR and SNAIL during human colon cancer progression. As indicated, downregulation of VDR has been found in some studies only in high-grade carcinomas, whereas in other studies it is observed already in low- and intermediate-grade tumours. These data indicate that the efficacy of therapy with 1,25(OH)2D3 analogues is lost at late steps of tumour progression.
These data suggest that VDR-deficient mice (whose phenotype closely resembles that of human with vitamin D-dependent rickets type II) may be prone to develop colon cancer. However, these animals display hyperproliferation and oxidative stress in the distal colon, but not elevated rates of spontaneous colon cancer (Kállay et al, 2001). Likewise, mice lacking 25-hydroxyvitamin D3-1α-hydroxylase show similar abnormalities to humans with vitamin D-dependent rickets type I, while no changes in intestines or tumour development have been reported (Bikle et al, 2004).
Several VDR polymorphisms have been described. However, most VDR polymorphisms do not result in structural alteration in the VDR protein and are of unknown functional significance and unrelated to adenoma recurrence, and furthermore do not modify the associations with vitamin D or calcium (Peters et al, 2001; Grau et al, 2003; Sutton and MacDonald, 2003; Harris and Go, 2004).
SNAIL: biological effects and regulation
SNAIL is a zinc-finger transcription factor expressed in migratory processes during embryonic development that has recently been implicated in cancer (Nieto, 2002). SNAIL upregulation in epithelial cells induces the acquisition of cell movement and invasiveness, at least in part due to the repression of E-cadherin expression (Batlle et al, 2000; Cano et al, 2000). In the E-cadherin gene promoter, SNAIL binds to regions that contain the consensus core sequence (CAGGTC), a motif that is a subset of the E-box (CANNTG), the conserved binding site of basic helix-loop-helix transcription factors. The acquisition of the invasive phenotype is linked to SNAIL upregulation and concomitant E-cadherin repression in gastric, melanoma, breast and hepatocellular cancer cells. Either directly or indirectly, SNAIL also downregulates other genes encoding adhesion proteins such as several claudins, occludin and ZO-1, and also those for MUC-1 and aromatase, while it upregulates several metalloproteinases in different cell types. Additionally, SNAIL attenuates cell proliferation and confers resistance to apoptosis in breast cancer cells (Vega et al, 2004). Supporting a role of SNAIL in colon tumorigenesis, downregulation of SNAIL suppresses both total tumour number and incidence of large tumours in APCmin mice (Roy et al, 2004). Studies performed with human and murine SNAIL gene promoters have evidenced the dependence of the transcription of this gene on the activity of signalling pathways involving MEK1/2, PI3K/Akt and NFκB/p65 (Peinado et al, 2003; Barberá et al, 2004). In mammary epithelial cells, SNAIL is repressed by MTA-3, a gene induced by oestrogen.
Relation between SNAIL and VDR expression
Our group observed that in a panel of human colon cancer cell lines, high SNAIL expression associates with undifferentiation and low VDR and E-cadherin expression, and vice versa. Subsequent work revealed that SNAIL protein binds to and represses (50%) the human VDR gene promoter (Pálmer et al, 2004). As a result, SNAIL abolishes the induction by 1,25(OH)2D3 of E-cadherin and other target genes (Figure 1). Moreover, SNAIL-overexpressing human colon cancer cells became resistant to growth inhibition by the vitamin D analogue EB1089 when they were injected subcutaneously in immune-deficient mice. In humans, we have found increased SNAIL RNA expression in tumour vs normal tissue (22 out of 32 patients), and a significant inverse correlation between the expression of SNAIL and that of VDR and E-cadherin (Pálmer et al, 2004). A later study of a larger series of patients confirmed this relation, and showed that loss of VDR RNA is linked to poor differentiation (F Bonilla, personal communication). These data indicate that the loss of VDR RNA expression during colon cancer progression is probably related to SNAIL upregulation, and therefore that high levels of SNAIL may be responsible for the failure of therapy with vitamin D analogues in patients with this and perhaps other neoplasias (Figure 2). In contrast to our data, SNAIL RNA expression was only detected in a low proportion of human colon tumours in another study (Rosivatz et al, 2004). The reasons for this discrepancy are unclear.
Loss of E-cadherin has been causally associated to the transition of adenoma to carcinoma and the acquisition of migration capacity. As stated above, VDR is also lost at late stages of colon cancer progression, impairing the induction of E-cadherin by 1,25(OH)2D3. Concordantly with these and our results, we propose that SNAIL promotes the acquisition of invasive behaviour via the repression of both VDR and E-cadherin genes. The repression of VDR by SNAIL is particularly relevant due to the protective action of 1,25(OH)2D3 and analogues on colon carcinogenesis, and constitutes a second mechanism for the efficient repression of E-cadherin. We have not found regulation of SNAIL RNA expression in colon cancer cells by 1,25(OH)2D3. Subcellular localisation and degradation of SNAIL protein is regulated by phosphorylation (Zhou et al, 2004) but, likewise, 1,25(OH)2D3 does not change SNAIL protein localisation in SW480-ADH cells.
During development, SNAIL expression is linked to cell deadhesion and migration processes, but it is downregulated when the cells have reached their destination. In human breast cancer metastasis E-cadherin is re-expressed (Bukholm et al, 2000), suggesting that SNAIL might be downregulated. Whether this occurs in colon metastasis remains to be elucidated. Likewise, no data are available on VDR expression in metastasis. The possibility that SNAIL upregulation reported in melanoma, gastric or other cancers may also cause VDR downregulation in these neoplasias remains unexplored. The VDR gene is subjected to complex transcriptional regulation: Wilm's tumour suppressor, Zeb-1, Cdx-2 and Sp1 transcription factors as well as protein kinase A induce VDR expression. At least during tumorigenesis, SNAIL seems to overcome these factors.
Therapeutic implication of the repression of VDR by SNAIL
Only a subset of cancer patients treated with vitamin D analogues respond to therapy. As with other therapies, it is important to select those patients that are likely to respond. An example is the selection of breast cancer patients with high HER2/Neu expression as candidates for treatment with anti-HER2/Neu antibodies. Our data indicate that colon cancer patients with SNAIL upregulation express low VDR and are thus likely to be poor responders to vitamin D analogues. As SNAIL upregulation seems to be linked to the acquisition of the migratory/invasive phenotype, which correlates with the metastatic potential, patients with low-grade colon cancers are preferential candidates for this therapy (Figure 2). In cases of acceptable toxicities, they may also be useful as chemopreventive agents for high-risk patients. Our studies show that the balance between VDR and SNAIL expression plays an important role in the control of cell fate during colon cancer progression, and emphasise the need to analyse VDR and/or SNAIL expression in colon cancers in order to select patients suitable for therapy with vitamin D analogues. An attractive, albeit unexplored, possibility is the study of SNAIL or VDR RNA levels in blood, a strategy used for cytokeratin 19, carcinoembryonic antigen and β-catenin in colorectal cancer (Silva et al, 2002) and other genes in different neoplasias. If upregulation of SNAIL or downregulation of VDR in tumours could be monitored by RT–PCR analysis of circulating RNA levels, this would constitute a valuable tool for the selection of candidates for therapy with vitamin D analogues.
Conclusions
1,25(OH)2D3 has profound effects on gene expression, inducing differentiation and inhibiting proliferation of colon cancer cells. Preclinical and epidemiological data suggest that vitamin D analogues may be useful for the prevention and treatment of colorectal cancer. To date, however, therapy with these compounds has benefited only a limited number of cancer patients. The SNAIL transcription factor inhibits the expression of genes encoding adhesive proteins and causes the acquisition of migratory and invasive phenotype of epithelial cells, and is thus considered an important factor of malignancy. Recent data have shown that SNAIL represses VDR gene expression in cultured human colon cancer cells, leading to hormone unresponsiveness in vitro and in vivo. Since SNAIL is upregulated during human colon cancer progression, which is associated with reduced VDR expression, the analysis of SNAIL expression may help to select patients suitable for therapy with vitamin D analogues.
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Barberá MJ, Puig I, Domínguez D, Julien-Grille S, Guaita-Esteruelas S, Peiró S, Baulida J, Franci C, Dedhar S, Larue L, García de Herreros A (2004) Regulation of Snail transcription during epithelial to mesenchymal transition of tumor cells. Oncogene 23: 7345–7354
Batlle E, Sancho E, Franci C, Domínguez D, Monfar M, Baulida J, García de Herreros A (2000) The transcription factor Snail is a repressor of E-cadherin gene expression in epithelial tumour cells. Nat Cell Biol 2: 84–89
Bikle DD, Chang S, Crumrine D, Elalieh H, Man MQ, Dardenne O, Xie Z, Arnaud RS, Feingold K, Elias PM (2004) Mice lacking 25(OH)D 1-α-hydroxylase demonstrate decreased epidermal differentiation and barrier function. J Steroid Biochem Mol Biol 89–90: 347–353
Bukholm IK, Neslandf JM, Borresen-Dale AL (2000) Re-expression of E-cadherin, α-catenin and β-catenin, but not of γ-catenin, in metastatic tissue from breast cancer patients. J Pathol 190: 3–5
Cano A, Pérez-Moreno MA, Rodrigo I, Locascio A, Blanco MJ, del Barrio MG, Portillo F, Nieto MA (2000) The transcription factor Snail controls epithelial–mesenchymal transitions by repressing E-cadherin expression. Nat Cell Biol 2: 76–83
Carlberg C, Mouriño A (2003) New vitamin D receptor ligands. Expert Opin Ther Patents 13: 761–772
Cross HS, Bajna E, Bises G, Gense D, Kállay E, Pötzi R, Wenzl E, Wrba F, Roka R, Peterlik M (1996) Vitamin D receptor and cytokeratin expression may be progression indicators in human colon cancer. Anticancer Res 16: 2333–2338
Cross HS, Bareis P, Hofer H, Bischof MG, Bajna E, Kriwanek S, Bonner E, Peterlik M (2001) 25-Hydroxyvitamin D3-1α-hydroxylase and vitamin D receptor gene expression in human colonic mucosa is elevated during early cancerogenesis. Steroids 66: 287–292
Evans SRT, Nolla J, Hanfelt J, Shabahang M, Nauta RJ, Shchepotin IB (1998) Vitamin D receptor expression as a predictive marker of biological behaviour in human colorectal cancer. Clin Cancer Res 4: 1591–1595
Ferlay J, Bray F, Sankila R, Parkin DM (1999) EUCAN: Cancer Incidence, Mortality and Prevalence in the European Union 1998, version 5.0. IARC Cancer Base. No. 4. Lyon: IARC Press
Giles RH, van Es JH, Clevers H (2003) Caught up in a Wnt storm: Wnt signalling in cancer. Biochim Biophys Acta 1653: 1–24
Grau MV, Baron JA, Sandler RS, Haile RW, Beach ML, Church TR, Heber D (2003) Vitamin D, calcium supplementation, and colorectal adenomas: results of a randomised trial. J Nat Cancer Inst 95: 1765–1771
Hansen CM, Binderup L, Hamberg KJ, Carlberg C (2001) Vitamin D and cancer: effects of 1,25(OH)2D3 and its analogs on growth control and tumorigenesis. Front Biosci 6: D820–D848
Harris DM, Go VLW (2004) Vitamin D and colon carcinogenesis. J Nutr 134: 3463S–3471S
Kállay E, Bareis P, Bajna E, Kriwanek S, Bonner E, Toyokuni S, Cross HS (2002) Vitamin D receptor activity and prevention of colonic hyperproliferation and oxidative stress. Food Chem Toxicol 40: 1191–1196
Kállay E, Pietschman P, Toyokuni S, Bajna E, Hahn P, Mazzucco K, Bieglmayer C, Kato S, Cross HS (2001) Characterization of a vitamin D receptor knockout mouse as a model of colorectal hyperproliferation and DNA damage. Carcinogenesis 22: 1429–1435
Lamprecht SA, Lipkin M (2003) Chemoprevention of colon cancer by calcium, vitamin D and folate: molecular mechanisms. Nat Rev Cancer 3: 601–614
MacDonald PN, Kraichely DM, Brown AJ (2001) The vitamin D receptor. In Nuclear Receptors and Genetic Disease Burris TP, McCabe ERB (eds) pp 197–243. London: Academic Press
Nieto MA (2002) The Snail superfamily of zinc-finger transcription factors. Nat Rev Cell Biol 3: 155–166
Pálmer HG, González-Sancho JM, Espada J, Berciano MT, Puig I, Baulida J, Quintanilla M, Cano A, García de Herreros A, Lafarga M, Muñoz A (2001) Vitamin D3 promotes the differentiation of colon carcinoma cells by the induction of E-cadherin and the inhibition of β-catenin signalling. J Cell Biol 154: 369–388
Pálmer HG, Larriba MJ, García JM, Ordóñez-Morán P, Peña C, Peiró S, Puig I, Rodríguez R, de la Fuente R, Bernad A, Pollán M, Bonilla F, Gamallo C, García de Herreros A, Muñoz A (2004) The transcription factor SNAIL represses vitamin D receptor expression and responsiveness in human colon cancer. Nat Med 10: 917–919
Pálmer HG, Sánchez-Carbayo M, Ordóñez-Morán P, Larriba MJ, Cordón-Cardó C, Muñoz A (2003) Genetic signatures of differentiation induced by 1α,25-dihydroxyvitamin D3 in human colon cancer cells. Cancer Res 63: 7799–7806
Peinado H, Quintanilla M, Cano A (2003) Transforming growth factor β-1 induces Snail transcription factor in epithelial cell lines. J Biol Chem 278: 21113–21123
Peters U, McGlynn KA, Chatterjee N, Gunter E, García-Closas M, Rothman N, Sinba R (2001) Vitamin D, calcium, and vitamin D receptor polymorphism in colorectal adenomas. Cancer Epidem Biomark 10: 1267–1274
Posner GH, Crawford KR, Yang HW, Kahraman M, Jeon HB, Li H, Lee JK, Suh BC, Hatcher MA, Labonte T, Usera A, Dolan PM, Kensler TW, Peleg S, Jones G, Zhang A, Korczak B, Saha U, Chuang SS (2004) Potent, low-calcemic, selective inhibitors of CYP24 hydroxylase: 24-sulfone analogs of the hormone 1α,25-dihydroxyvitamin D3 . J Steroid Biochem Mol Biol 89–90: 5–12
Prüfer K, Barsony J (2002) Retinoid X receptor dominates the nuclear import and export of the unliganded vitamin D receptor. Mol Endocrinol 16: 1738–1751
Rosivatz E, Becker I, Specht K, Fricke E, Luber B, Busch R, Höfler H, Becker K-F (2004) Neoexpression of N-cadherin in E-cadherin positive colon cancers. Int J Cancer 111: 711–719
Roy HK, Iversen P, Hart J, Liu Y, Koetsier JL, Kim Y, Kunte DP, Madugula M, Backman V, Wali RK (2004) Down-regulation of SNAIL suppresses MIN mouse tumorigenesis: modulation of apoptosis, proliferation, and fractal dimension. Mol Cancer Ther 3: 1159–1165
Sheinin Y, Kaserer K, Wrba F, Wenzl E, Kriwanek S, Peterlink M, Cross HS (2000) In situ mRNA hybridization analysis and immunolocalization of the vitamin D receptor in normal and carcinomatous human colonic mucosa: relation to epidermal growth factor receptor expression. Virchows Arch 437: 501–507
Silva JM, Rodríguez R, García JM, Muñoz C, Silva J, Domínguez G, Provencio M, España P, Bonilla F (2002) Detection of epithelial tumor RNA in the plasma of colon cancer patients is associated with advanced stages. Gut 50: 530–534
Sutton ALM, MacDonald PN (2003) Vitamin D: more than a ‘bone-a-fide’ hormone. Mol Endocrinol 17: 777–791
Vega S, Morales AV, Ocaña OH, Valdés F, Fabregat I, Nieto MA (2004) Snail blocks the cell cycle and confers resistance to cell death. Gene Dev 18: 1131–1143
Zhou BP, Deng J, Xia W, Xu J, Li YM, Gunduz M, Hung M-C (2004) Dual regulation of Snail by GSK-3β-mediated phosphorylation in control of epithelial–mesenchymal transition. Nat Cell Biol 6: 931–940
Acknowledgements
We apologise to the many investigators whose primary research publications could not be cited because of space constraints. We thank Drs Carsten Carlberg and Antonio García de Herreros for critically reading the manuscript, Drs Angela Nieto, José Miguel García, Cristina Peña and Marina Pollán for comments, Dr Félix Bonilla for communicating unpublished results and Robin Rycroft for his valuable assistance in the preparation of the English manuscript. The work of the authors on this topic is supported by Grants from Fundación Científica de la Asociación Española contra el Cáncer, Fundación de Investigación Médica Mutua Madrileña, Instituto de Salud Carlos III (FIS03-C03/10) and Ministerio de Educación y Ciencia (SAF04-1015) of Spain.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Larriba, M., Muñoz, A. SNAIL vs vitamin D receptor expression in colon cancer: therapeutics implications. Br J Cancer 92, 985–989 (2005). https://doi.org/10.1038/sj.bjc.6602484
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bjc.6602484
Keywords
This article is cited by
-
The role of vitamin D in reducing cancer risk and progression
Nature Reviews Cancer (2014)