Abstract
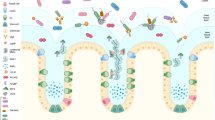
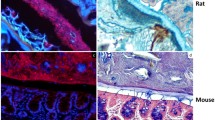
In discussions on intestinal protection, the protective capacity of mucus has not been very much considered. The progress in the last years in understanding the molecular nature of mucins, the main building blocks of mucus, has, however, changed this. The intestinal enterocytes have their apical surfaces covered by transmembrane mucins and the whole intestinal surface is further covered by mucus, built around the gel-forming mucin MUC2. The mucus of the small intestine has only one layer, whereas the large intestine has a two-layered mucus where the inner, attached layer has a protective function for the intestine, as it is impermeable to the luminal bacteria.






Similar content being viewed by others
References
Johansson ME, Phillipson M, Petersson J, Velcich A, Holm L, Hansson GC (2008) The inner of the two Muc2 mucin-dependent mucus layers in colon is devoid of bacteria. Proc Natl Acad Sci USA 105:15064–15069
Gill N, Wlodarska M, Finlay BB (2010) The future of mucosal immunology: studying an integrated system-wide organ. Nat Immunol 11:558–560
Lang T, Hansson GC, Samuelsson T (2007) Gel-forming mucins appeared early in metazoan evolution. Proc Natl Acad Sci USA 104:16209–16214
Lang T, Alexandersson M, Hansson GC, Samuelsson T (2004) Bioinformatic identification of polymerizing and transmembrane mucins in the puffer fish Fugu rubripes. Glycobiology 14:521–527
Vinall LE, King M, Novelli M, Green CA, Daniels G, Hilkens J, Sarner M, Swallow DM (2002) Altered expression and allelic association of the hypervariable membrane mucin MUC1 in Helicobacter pylori gastritis. Gastroenterology 123:41–49
Axelsson MA, Karlsson NG, Steel DM, Ouwendijk J, Nilsson T, Hansson GC (2001) Neutralization of pH in the Golgi apparatus causes redistribution of glycosyltransferases and changes in the O-glycosylation of mucins. Glycobiology 11:633–644
Olson FJ, Johansson ME, Klinga-Levan K, Bouhours D, Enerbäck L, Hansson GC, Karlsson NG (2002) Blood group A glycosyltransferase occurring as alleles with high sequence difference is transiently induced during a Nippostrongylus brasiliensis parasite infection. J Biol Chem 277:15044–15052
Holmén JM, Olson FJ, Karlsson H, Hansson GC (2002) Two glycosylation alterations of mouse intestinal mucins due to infection caused by the parasite Nippostrongylus brasiliensis. Glycoconj J 19:67–75
Atuma C, Strugala V, Allen A, Holm L (2001) The adherent gastrointestinal mucus gel layer: thickness and physical state in vivo. Am J Physiol Gastrointest Liver Physiol 280:G922–G929
Sheng YH, Lourie R, Linden SK, Jeffery PL, Roche D, Tran TV, Png CW, Waterhouse N, Sutton P, Florin TH, McGuckin MA (2011) The MUC13 cell-surface mucin protects against intestinal inflammation by inhibiting epithelial cell apoptosis. Gut. on-line
Carlstedt I, Herrmann A, Karlsson H, Sheehan J, Fransson LA, Hansson GC (1993) Characterization of two different glycosylated domains from the insoluble mucin complex of rat small intestine. J Biol Chem 268:18771–18781
Macao B, Johansson DG, Hansson GC, Härd T (2006) Autoproteolysis coupled to protein folding in the SEA domain of the membrane-bound MUC1 mucin. Nat Struct Mol Biol 13:71–76
Malmberg EK, Pelaseyed T, Petersson AC, Seidler UE, De Jonge H, Riordan JR, Hansson GC (2008) The C-terminus of the transmembrane mucin MUC17 binds to the scaffold protein PDZK1 that stably localizes it to the enterocyte apical membrane in the small intestine. Biochem J 410:283–289
Pelaseyed T, Hansson GC (2011) CFTR anion channel modulates expression of human transmembrane mucin MUC3 via the PDZ protein GOPC. J Cell Sci 124:3074–3083
Lamprecht G, Seidler U (2006) The emerging role of PDZ adapter proteins for regulation of intestinal ion transport. Am J Physiol Gastrointest Liver Physiol 291:G766–G777
Park SW, Zhen G, Verhaeghe C, Nakagami Y, Nguyenvu LT, Barczak AJ, Killeen N, Erle DJ (2009) The protein disulfide isomerase AGR2 is essential for production of intestinal mucus. Proc Natl Acad Sci USA 106:6950–6955
Kaser A, Lee AH, Franke A, Glickman JN, Zeissig S, Tilg H, Nieuwenhuis EE, Higgins DE, Schreiber S, Glimcher LH, Blumberg RS (2008) XBP1 links ER stress to intestinal inflammation and confers genetic risk for human inflammatory bowel disease. Cell 134:743–756
Godl K, Johansson ME, Lidell ME, Mörgelin M, Karlsson H, Olson FJ, Gum JR Jr, Kim YS, Hansson GC (2002) The N terminus of the MUC2 mucin forms trimers that are held together within a trypsin-resistant core fragment. J Biol Chem 277:47248–47256
Herrmann A, Davies JR, Lindell G, Martensson S, Packer NH, Swallow DM, Carlstedt I (1999) Studies on the “insoluble” glycoprotein complex from human colon. Identification of reduction-insensitive MUC2 oligomers and C-terminal cleavage. J Biol Chem 274:15828–15836
Axelsson MA, Asker N, Hansson GC (1998) O-glycosylated MUC2 monomer and dimer from LS 174T cells are water-soluble, whereas larger MUC2 species formed early during biosynthesis are insoluble and contain nonreducible intermolecular bonds. J Biol Chem 273:18864–18870
Ambort D, van der Post S, Johansson ME, Mackenzie J, Thomsson E, Krengel U, Hansson G (2011) Function of the CysD domain of the gel-forming MUC2 mucin. Biochem J 436:61–70
Johansson ME, Thomsson KA, Hansson GC (2009) Proteomic analyses of the two mucus layers of the colon barrier reveal that their main component, the Muc2 mucin, is strongly bound to the Fcgbp protein. J Proteome Res 8:3549–3557
Lidell ME, Johansson ME, Hansson GC (2003) An autocatalytic cleavage in the C terminus of the human MUC2 mucin occurs at the low pH of the late secretory pathway. J Biol Chem 278:13944–13951
Zhuo L, Hascall VC, Kimata K (2004) Inter-alpha-trypsin inhibitor, a covalent protein–glycosaminoglycan–protein complex. J Biol Chem 279:38079–38082
Johansson ME, Larsson JM, Hansson GC (2011) The two mucus layers of colon are organized by the MUC2 mucin, whereas the outer layer is a legislator of host-microbial interactions. Proc Natl Acad Sci USA 108:4659–4665
Subramani DB, Johansson MEV, Dahlén G, Hansson GC (2010) Lactobacillus and Bifidobacterium species do not secrete proteases that can cleave the MUC2 mucin protein core which organizes the colon mucus layers. Beneficial Microbes 1:343–350
Velcich A, Yang W, Heyer J, Fragale A, Nicholas C, Viani S, Kucherlapati R, Lipkin M, Yang K, Augenlicht L (2002) Colorectal cancer in mice genetically deficient in the mucin Muc2. Science 295:1726–1729
Van der Sluis M, De Koning BA, De Bruijn AC, Velcich A, Meijerink JP, Van Goudoever JB, Buller HA, Dekker J, Van Seuningen I, Renes IB, Einerhand AW (2006) Muc2-deficient mice spontaneously develop colitis, indicating that MUC2 is critical for colonic protection. Gastroenterology 131:117–129
Ley RE, Bäckhed F, Turnbaugh P, Lozupone CA, Knight RD, Gordon JI (2005) Obesity alters gut microbial ecology. Proc Natl Acad Sci USA 102:11070–11075
Gagneux P, Varki A (1999) Evolutionary considerations in relating oligosaccharide diversity to biological function. Glycobiology 9:747–755
Hansson GC (1988) Structural aspects of blood group glycosphingolipids in the gastrointestinal tract. Adv Exp Med Biol 228:465–494
Larsson JM, Karlsson H, Sjovall H, Hansson GC (2009) A complex, but uniform O-glycosylation of the human MUC2 mucin from colonic biopsies analyzed by nanoLC/MSn. Glycobiology 19:756–766
Larsson JM, Karlsson H, Crespo JG, Johansson ME, Eklund L, Sjövall H, Hansson GC (2011) Altered O-glycosylation profile of MUC2 mucin occurs in active ulcerative colitis and is associated with increased inflammation. Inflamm Bowel Dis. on-line
Xu J, Bjursell MK, Himrod J, Deng S, Carmichael LK, Chiang HC, Hooper LV, Gordon JI (2003) A genomic view of the human–Bacteroides thetaiotaomicron symbiosis. Science 299:2074–2076
Fu J, Wei B, Wen T, Johansson ME, Liu X, Bradford E, Thomsson KA, McGee S, Mansour L, Tong M, McDaniel JM, Sferra TJ, Turner JR, Chen H, Hansson GC, Braun J, Xia L (2011) Loss of intestinal core 1-derived O-glycans causes spontaneous colitis in mice. J Clin Invest 121:1657–1666
Acknowledgments
This work was supported by the Swedish Research Council (no. 7461, 21027, and 342-2004-4434), The Swedish Cancer Foundation, The Knut and Alice Wallenberg Foundation (KAW2007.0118), IngaBritt and Arne Lundberg Foundation, Sahlgren’s University Hospital (LUA-ALF), EU-FP7 IBDase (no. 200931), Wilhelm and Martina Lundgren’s Foundation, Torsten och Ragnar Söderbergs Stiftelser, The Sahlgrenska Academy Foundations, and The Swedish Foundation for Strategic Research—The Mucosal Immunobiology and Vaccine Center (MIVAC) and the Mucus-Bacteria-Colitis Center (MBC) of the Innate Immunity Program (2010-2014). We also acknowledge the Mammalian Protein Expression Core Facility at the University of Gothenburg.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Johansson, M.E.V., Ambort, D., Pelaseyed, T. et al. Composition and functional role of the mucus layers in the intestine. Cell. Mol. Life Sci. 68, 3635–3641 (2011). https://doi.org/10.1007/s00018-011-0822-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-011-0822-3