Abstract
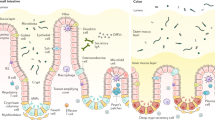
Recent data indicate that stimulation of Toll-like receptors (TLRs) by intestinal microbiota supports colonic homeostasis. Intestinal epithelial cells (IECs) express several TLRs. It has been demonstrated that repeated exposure to TLR ligands causes IECs to become hypo-responsive. Inhibition of TLR cell surface expression and the induction of inhibitory intracellular signaling molecules have been proposed to mediate this state of tolerance to luminal commensals and/or their TLR ligands. In this review, we summarize our recent observations that uncover the essential role of IEC polarization in the homeostatic response of IECs to TLR ligands. In particular, apical TLR9 stimulation delivers negative signals that curtail inflammatory responses induced by basolateral stimulation by other TLRs. This “polarizing–tolerizing” ability of IECs represents a unique mechanism that restrains inflammatory responses in a bacteria-rich environment. Thus, interventions that enhance IEC polarization could prevent or inhibit human inflammatory bowel diseases and other types of intestinal inflammation.









Similar content being viewed by others
References
Akhtar M, Watson JL, Nazli A, McKay DM (2003) Bacterial DNA evokes epithelial IL-8 production by a MAPK-dependent, NFkappaB-independent pathway. FASEB J 17:1319–1321
Akira S, Hemmi H (2003) Recognition of pathogen-associated molecular patterns by TLR family. Immunol Lett 85:85–95
Balda MS, Whitney JA, Flores C, Gonzalez S, Cereijido M, Matter K (1996) Functional dissociation of paracellular permeability and transepithelial electrical resistance and disruption of the apical–basolateral intramembrane diffusion barrier by expression of a mutant tight junction membrane protein. J Cell Biol 134:1031–1049
Bantel H, Schmitz ML, Raible A, Gregor M, Schulze-Osthoff K (2002) Critical role of NF-kappaB and stress-activated protein kinases in steroid unresponsiveness. FASEB J 16:1832–1834
Bauer S, Kirschning CJ, Hacker H, Redecke V, Hausmann S, Akira S, Wagner H, Lipford GB (2001) Human TLR9 confers responsiveness to bacterial DNA via species-specific CpG motif recognition. Proc Natl Acad Sci USA 98:9237–9242
Beinke S, Robinson MJ, Hugunin M, Ley SC (2004) Lipopolysaccharide activation of the TPL-2/MEK/extracellular signal-regulated kinase mitogen-activated protein kinase cascade is regulated by IkappaB kinase-induced proteolysis of NF-kappaB1 p105. Mol Cell Biol 24:9658–9667
Cario E, Brown D, McKee M, Lynch-Devaney K, Gerken G, Podolsky DK (2002) Commensal-associated molecular patterns induce selective toll-like receptor-trafficking from apical membrane to cytoplasmic compartments in polarized intestinal epithelium. Am J Pathol 160:165–173
Cario E, Podolsky DK (2005) Intestinal epithelial TOLLerance versus inTOLLerance of commensals. Mol Immunol 42:887–893
Cario E, Gerken G, Podolsky DK (2007) Toll-like receptor 2 controls mucosal inflammation by regulating epithelial barrier function. Gastroenterology 132:1359–1374
Cohen S, Achbert-Weiner H, Ciechanover A (2004) Dual effects of IkappaB kinase beta-mediated phosphorylation on p105 Fate: SCF(beta-TrCP)-dependent degradation and SCF(beta-TrCP)-independent processing. Mol Cell Biol 24:475–486
Cooper HS, Murthy SN, Shah RS, Sedergran DJ (1993) Clinicopathologic study of dextran sulfate sodium experimental murine colitis. Lab Invest 69:238–249
Farquhar MG, Palade GE (1963) Junctional complexes in various epithelia. J Cell Biol 17:375–412
Gallucci S, Matzinger P (2001) Danger signals: SOS to the immune system. Curr Opin Immunol 13:114–119
Gewirtz AT, Navas TA, Lyons S, Godowski PJ, Madara JL (2001) Cutting edge: bacterial flagellin activates basolaterally expressed TLR5 to induce epithelial proinflammatory gene expression. J Immunol 167:1882–1885
Lee J, Mo J-H, Katakura K, Alkalay I, Rucker AN, Liu Y-T, Lee H-K, Shen C, Cojocaru G, Shenouda S, Kagnoff M, Eckmann L, Ben-Neriah Y, Raz E (2006) Maintenance of colonic homeostasis by distinctive apical TLR9 signaling in intestinal epithelial cells. Nat Cell Biol 8(12):1327–1336
Karin M, Ben-Neriah Y (2000) Phosphorylation meets ubiquitination: the control of NF-[kappa]B activity. Annu Rev Immunol 18:621–663
Kazmierczak BI, Mostov K, Engel JN (2001) Interaction of bacterial pathogens with polarized epithelium. Annu Rev Microbiol 55:407–435
Kelly D, Campbell JI, King TP, Grant G, Jansson EA, Coutts AG, Pettersson S, Conway S (2004) Commensal anaerobic gut bacteria attenuate inflammation by regulating nuclear-cytoplasmic shuttling of PPAR-gamma and RelA. Nat Immunol 5:104–112
Macfarlane DE, Manzel L (1998) Antagonism of immunostimulatory CpG-oligodeoxynucleotides by quinacrine, chloroquine, and structurally related compounds. J Immunol 160:1122–1131
Marrero JA, Matkowskyj KA, Yung K, Hecht G, Benya RV (2000) Dextran sulfate sodium-induced murine colitis activates NF-kappaB and increases galanin-1 receptor expression. Am J Physiol Gastrointest Liver Physiol 278:G797–G804
Mowat AM (2003) Anatomical basis of tolerance and immunity to intestinal antigens. Nat Rev Immunol 3:331–341
Naidu AS, Bidlack WR, Clemens RA (1999) Probiotic spectra of lactic acid bacteria (LAB). Crit Rev Food Sci Nutr 39:13–126
Ortega-Cava CF, Ishihara S, Rumi MA, Kawashima K, Ishimura N, Kazumori H, Udagawa J, Kadowaki Y, Kinoshita Y (2003) Strategic compartmentalization of Toll-like receptor 4 in the mouse gut. J Immunol 170:3977–3985
Otte JM, Cario E, Podolsky DK (2004) Mechanisms of cross hyporesponsiveness to Toll-like receptor bacterial ligands in intestinal epithelial cells. Gastroenterology 126:1054–1070
Pedersen G, Andresen L, Matthiessen MW, Rask-Madsen J, Brynskov J (2005) Expression of Toll-like receptor 9 and response to bacterial CpG oligodeoxynucleotides in human intestinal epithelium. Clin Exp Immunol 141:298–306
Schmausser B, Andrulis M, Endrich S, Lee SK, Josenhans C, Muller-Hermelink HK, Eck M (2004) Expression and subcellular distribution of toll-like receptors TLR4, TLR5 and TLR9 on the gastric epithelium in Helicobacter pylori infection. Clin Exp Immunol 136:521–526
Takeda K, Kaisho T, Akira S (2003) Toll-like receptors. Annu Rev Immunol 21:335–376
van Es JH, Jay P, Gregorieff A, van Gijn ME, Jonkheer S, Hatzis P, Thiele A, van den Born M, Begthel H, Brabletz T, Taketo MM, Clevers H (2005) Wnt signalling induces maturation of Paneth cells in intestinal crypts. Nat Cell Biol 7:381–386
Van ISC, Maier O, Van Der Wouden JM, Hoekstra D (2000) The subapical compartment and its role in intracellular trafficking and cell polarity. J Cell Physiol 184:151–160
Waterfield MR, Zhang M, Norman LP, Sun SC (2003) NF-kappaB1/p105 regulates lipopolysaccharide-stimulated MAP kinase signaling by governing the stability and function of the Tpl2 kinase. Mol Cell 11:685–694
Yi AK, Tuetken R, Redford T, Waldschmidt M, Kirsch J, Krieg AM (1998) CpG motifs in bacterial DNA activate leukocytes through the pH-dependent generation of reactive oxygen species. J Immunol 160:4755–4761
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by NIH grants AI68685, AI40682, DK35108, and the CCFA.
Rights and permissions
About this article
Cite this article
Lee, J., Gonzales-Navajas, J.M. & Raz, E. The “polarizing–tolerizing” mechanism of intestinal epithelium: its relevance to colonic homeostasis. Semin Immunopathol 30, 3–9 (2008). https://doi.org/10.1007/s00281-007-0099-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00281-007-0099-7