Abstract
Background
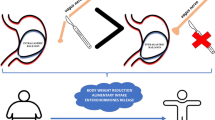
The aim of this study was to evaluate whether gastric bypass with or without vagal preservation resulted in a different outcome.
Methods
Body weight, food intake and postprandial peptide YY (PYY) and glucagon-like peptide (GLP-1) levels were compared between gastric bypass (n = 55) and sham-operated rats (n = 27) in three groups. In group 1 (n = 17), the vagal nerve was not preserved, while in group 2 the vagal nerve was preserved during gastric bypass (n = 10). In group 3, gastric bypass rats (n = 28) were randomised for either one of the two techniques.
Results
Rats in which the vagal nerve was preserved during gastric bypass showed a lower body weight (p < 0.001) and reduced food intake (p < 0.001) compared to rats in which the vagal nerve was not preserved during the gastric bypass operation. Levels of PYY and GLP-1 were significantly increased after gastric bypass compared to sham-operated controls (p < 0.05), but there was no difference between gastric bypass rats with and without vagal preservation. Differences in food intake and body weight were not related to the size of the gastro-jejunostomy in gastric bypass rats. There were no signs of malabsorption or inflammation after gastric bypass.
Conclusion
We propose that the vagal nerve should be preserved during the gastric bypass operation as this might play an important role for the mechanisms that induce weight loss and reduce food intake in rats. In contrast, the gastro-jejunal stoma size was found to be of minor relevance.



Similar content being viewed by others
References
Adams TD, Gress RE, Smith SC, et al. Long-term mortality after gastric bypass surgery. N Engl J Med. 2007;357:753–61.
Sjostrom L, Narbro K, Sjostrom CD, et al. Effects of bariatric surgery on mortality in Swedish obese subjects. N Engl J Med. 2007;357:741–52.
Buchwald H, Avidor Y, Braunwald E, et al. Bariatric surgery: a systematic review and meta-analysis. JAMA. 2004;292:1724–37.
Buchwald H. Bariatric surgery for morbid obesity: health implications for patients, health professionals, and third-party payers. J Am Coll Surg. 2005;200:593–604.
Borg CM, le Roux CW, Ghatei MA, et al. Progressive rise in gut hormone levels after Roux-en-Y gastric bypass suggests gut adaptation and explains altered satiety. Br J Surg. 2006;93:210–5.
Bueter M, Lowenstein C, Olbers T et al. Gastric bypass increases energy expenditure in rats. Gastroenterology 2010.
Guijarro A, Suzuki S, Chen C, et al. Characterization of weight loss and weight regain mechanisms after Roux-en-Y gastric bypass in rats. Am J Physiol Regul Integr Comp Physiol. 2007;293:R1474–89.
Furnes MW, Tommeras K, Arum CJ, et al. Gastric bypass surgery causes body weight loss without reducing food intake in rats. Obes Surg. 2008;18:415–22.
Furnes MW, Stenstrom B, Tommeras K, et al. Feeding behavior in rats subjected to gastrectomy or gastric bypass surgery. Eur Surg Res. 2008;40:279–88.
Guijarro A, Osei-Hyiaman D, Harvey-White J, et al. Sustained weight loss after Roux-en-Y gastric bypass is characterized by down regulation of endocannabinoids and mitochondrial function. Ann Surg. 2008;247:779–90.
Meguid MM, Glade MJ, Middleton FA. Weight regain after Roux-en-Y: a significant 20% complication related to PYY. Nutrition. 2008;24:832–42.
Stenstrom B, Furnes MW, Tommeras K, et al. Mechanism of gastric bypass-induced body weight loss: one-year follow-up after micro-gastric bypass in rats. J Gastrointest Surg. 2006;10:1384–91.
Stylopoulos N, Hoppin AG, Kaplan LM. Roux-en-Y gastric bypass enhances energy expenditure and extends lifespan in diet-induced obese rats. Obesity (Silver Spring). 2009;17:1839–47.
Tichansky DS, Boughter Jr JD, Harper J, et al. Gastric bypass surgery in rats produces weight loss modeling after human gastric bypass. Obes Surg. 2008;18:1246–50.
Wang Y, Liu J. Combination of bypassing stomach and vagus dissection in high-fat diet-induced obese rats-a long-term investigation. Obes Surg. 2010.
Perathoner A, Weiss H, Santner W, et al. Vagal nerve dissection during pouch formation in laparoscopic Roux-Y-gastric bypass for technical simplification: does it matter? Obes Surg. 2009;19:412–7.
Sundbom M, Holdstock C, Engstrom BE, et al. Early changes in ghrelin following Roux-en-Y gastric bypass: influence of vagal nerve functionality? Obes Surg. 2007;17:304–10.
Berthoud HR. The vagus nerve, food intake and obesity. Regul Pept. 2008;149:15–25.
Niederhausern W.v. Recherches experimentales sur la fasciculation et la terminaison des nerfs vagues dans l'abdomen et leurs rapports avec l'innervation renale chez le rat. C.R.Assoc.Anat. 40, 783–784. 1953. Ref Type: Generic
Jackson RJ, Davis WB, Macdonald I. The energy values of carbohydrates: should bomb calorimeter data be modified? Proc Nutr Soc. 1977;36:90A.
Korner J, Bessler M, Cirilo LJ, et al. Effects of Roux-en-Y gastric bypass surgery on fasting and postprandial concentrations of plasma ghrelin, peptide YY, and insulin. J Clin Endocrinol Metab. 2005;90:359–65.
le Roux CW, Aylwin SJ, Batterham RL, et al. Gut hormone profiles following bariatric surgery favor an anorectic state, facilitate weight loss, and improve metabolic parameters. Ann Surg. 2006;243:108–14.
Batterham RL, Cowley MA, Small CJ, et al. Gut hormone PYY(3-36) physiologically inhibits food intake. Nature. 2002;418:650–4.
Batterham RL, Cohen MA, Ellis SM, et al. Inhibition of food intake in obese subjects by peptide YY3-36. N Engl J Med. 2003;349:941–8.
Cone RD, Cowley MA, Butler AA, et al. The arcuate nucleus as a conduit for diverse signals relevant to energy homeostasis. Int J Obes Relat Metab Disord. 2001;25 Suppl 5:S63–7.
Larsen PJ, Tang-Christensen M, Jessop DS. Central administration of glucagon-like peptide-1 activates hypothalamic neuroendocrine neurons in the rat. Endocrinology. 1997;138:4445–55.
Abbott CR, Monteiro M, Small CJ, et al. The inhibitory effects of peripheral administration of peptide YY(3-36) and glucagon-like peptide-1 on food intake are attenuated by ablation of the vagal-brainstem-hypothalamic pathway. Brain Res. 2005;1044:127–31.
Muller MK, Rader S, Wildi S, et al. Long-term follow-up of proximal versus distal laparoscopic gastric bypass for morbid obesity. Br J Surg. 2008;95:1375–9.
Acknowledgements
M. B. was supported by the Deutsche Forschungsgemeinschaft (DFG). T.L. and C.L. were supported by the Swiss National Research Foundation. S.B. and C.le R. were supported by a Department of Health Clinician scientist award. Imperial College London receives support from the NIHR Biomedical Research Centre funding scheme.
Disclosure
The authors have no conflict of interest to disclose.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bueter, M., Löwenstein, C., Ashrafian, H. et al. Vagal Sparing Surgical Technique but Not Stoma Size Affects Body Weight Loss in Rodent Model of Gastric Bypass. OBES SURG 20, 616–622 (2010). https://doi.org/10.1007/s11695-010-0075-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-010-0075-5