Abstract
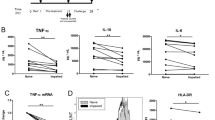
Neutralization of proinflammatory cytokines, such as tumor necrosis factor-α (TNF-α) or interleukin-1 (IL-1), decreases mortality in several animal models of sepsis. However, recent clinical trials did not show an unequivocal improvement in survival. In contrast to animals, which succumb to shock during the first 72 hours, we found that many patients die much later with signs of opportunistic infections accompanied by downregulation of their monocytic HLA-DR expression and reduced ability to produce lipopolysaccharide (LPS)-induced TNF-α in vitro1–3. This phenomenon of monocyte deactivation in septic patients with fatal outcome shows similarities to experimental monocytic refractoriness induced by LPS desensitization or by pretreatment with its endogenous mediators IL-10 and transforming growth factor-β (TGF-β)4. In order to strengthen their antimicrobial defense, here we tested whether interferon-γ (IFN-γ) can improve monocytic functions in these patients and in experimental monocytic deactivation. The considerably lowered in vitro levels of LPS-induced TNF-α in these situations were significantly enhanced by IFN-γ, but did not reach the extremely high levels of IFN-γ primed naive cells from healthy donors. Moreover, IFN-γ applied to septic patients with low monocytic HLA-DR expression restored the deficient HLA-DR expression and in vitro LPS-induced TNF-α secretion. Recovery of monocyte function resulted in clearance of sepsis in eight of nine patients. These data suggest that IFN-γ treatment in carefully selected septic patients is a novel therapeutic strategy worth pursuing.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Volk, H.D. et al. Diagnostic value of an immune monitoring program for the clinical management of immunosuppressed patients with septic complications. Clin. Transplant 3. 246–252 (1989).
Volk, H.D. et al. Alterations in function and phenotype of monocytes from patients with septic disease: Predictive value and new therapeutic strategies. in Host Defense Dysfunction in Trauma, Shock and Sepsis. (eds. Faist, E. and Meakins, J.L.) 246–271 (Springer-Verlag, Berlin-Heidelberg-New York, 1993).
Döcke, W.D. et al. Monocytic deactivation in fatal septic disease — Role of tumor necrosis factor-α and interleukin-10. in The Immune Consequences of Trauma, Shock and Sepsis — Mechanisms and Therapeutic Approaches. Vol.1, MOF, MODS and SIRS: Basic Mechanisms in Inflammation and Tissue Injury, (eds. Faist, E., Baue, A.E. & Schildberg, F.W.) 162–168 (Pabst Science Publishers, Lengerich, Germany, 1996).
Randow, F. Mechanism of endotoxin desensitization: Involvement of interleukin 10 and transforming growth factor beta. J. Exp. Med. 181, 1887–1892 (1995).
Beutler, B., Milsark, I.W. & Cerami, A.C. Passive immunization against cachectin/tumor necrosis factor protects mice from lethal effect of endotoxin. Science 229, 869–871 (1985).
Natanson, C., Hoffman, W.D., Suffredini, A.F., Eichacker, P.Q. & Danner, R.L. Selected treatment strategies for septic shock based on proposed mechanisms of pathogenesis. Ann. Intern. Med. 120, 771–783 (1994).
Fisher, C.J. et al. Recombinant human interleukin 1 receptor antagonist in the treatment of patients with sepsis syndrome: Results from a randomized, double-blind, placebo-controlled trial. JAMA 271, 1836–1843 (1994).
Fisher, C.J. et al. Treatment of septic shock with the tumor necrosis factor receptor:Fc fusion protein. N. Engl. J. Med. 334, 1697–1702 (1996).
Echtenacher, B., Falk, W., Mannel, D.N. & Krammer, P.H. Requirement of endogenous tumor necrosis factor/cachectin for recovery from experimental peritonitis. J. Immunol. 145, 3762–3766 (1990).
Pfeffer, K. et al. Mice deficient for the 55 kD tumor necrosis factor receptor are resistant to endotoxic shock, yet succumb to L. monocytogenes infection. Cell 73, 457–467 (1993).
Platzer, C., Meisel, C., Vogt, K., Platzer, M. & Volk, H.D. Up-regulation of mono-cytic IL-10 by tumor necrosis factor-alpha and cAMP elevating drugs. Int. Immunol. 7, 517–523 (1995).
Bone, R.C. Sir Isaac Newton, sepsis, and CARS. Crit. Care Med. 24, 1125–1128 (1996).
Munoz, C. et al. Dysregulation of in vitro cytokine production by monocytes during sepsis. J. Clin. Invest. 88, 1747–1754 (1991).
Faist, E. et al. Immunomodulatory therapy with thymopentin and indomethacin: Successful restoration of interleukin-2 synthesis in patients undergoing major surgery. Ann. Surg. 214, 264–273 (1991).
Young, H.A. & Hardy K.J. Role of interferon-gamma in immune cell regulation. J.Leukocyt. Biol. 58, 373–381 (1995).
Hart, P.H., Whitty G.A., Piccoli, D.S. & Hamilton, J.A. Control by IFN-gamma and PGE2 of TNF-alpha and IL-1 production by human monocytes. Immunology 66, 376–383 (1989).
Donnelly, R.P., Freeman, S.L. & Hayes, M.P. Inhibition of IL-10 expression by IFN-gamma up-regulates transcription of TNF-alpha in human monocytes. J. Immunol. 155, 1420–1427 (1995).
Heinzel, F.P. The role of IFN-gamma in the pathology of experimental endotoxemia. J. Immunol. 145, 2920–2924 (1990).
Bone, R.C. Definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. The ACCP/SCCM Consensus Conference Committee, American College of Chest Physicians/Society of Critical Care Medicine. Chest 101, 1644–1655 (1992).
Kaul, M., Eiselstein, J., Kempeni, J. & Daum, L. TNF-neutralization in sepsis guided by IL-6 serum levels. Sixth International TNF Congress, Rhodes, Hellas, Greece, 8–12 May 1996. [Abstr.] Eur. Cytokine Netw. 7, 283 (1996).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Döcke, WD., Randow, F., Syrbe, U. et al. Monocyte deactivation in septic patients: Restoration by IFN-γ treatment. Nat Med 3, 678–681 (1997). https://doi.org/10.1038/nm0697-678
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nm0697-678
This article is cited by
-
Immunosuppressive effects of circulating bile acids in human endotoxemia and septic shock: patients with liver failure are at risk
Critical Care (2023)
-
Interrogating the sepsis host immune response using cytomics
Critical Care (2023)
-
Mechanosensitive traction force generation is regulated by the neutrophil activation state
Scientific Reports (2023)
-
Adoptive macrophage directed photodynamic therapy of multidrug-resistant bacterial infection
Nature Communications (2023)
-
Prospective flow cytometry analysis of leucocyte subsets in critically ill patients who develop sepsis: a pilot study
Infection (2023)