Abstract
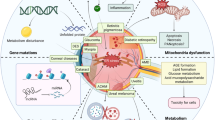
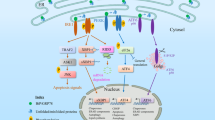
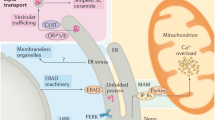
Endoplasmic reticulum (ER) stress is caused by the accumulation of unfolded proteins in the ER lumen, and is associated with vascular and neurodegenerative diseases. Although the connection between ER stress and some disease-related proteins has been studied using animal models of these diseases, no in vivo data concerning ER stress are available. Here we report a new method for monitoring ER stress in vivo, based on XBP-1 mRNA splicing by inositol requiring-1 (IRE-1) during ER stress. The stress indicator was constructed by fusing XBP-1 and venus, a variant of green fluorescent protein. During stress, the spliced indicator mRNA is translated into an XBP-1-venus fusion protein, which can be detected by its fluorescence. We used transgenic animals expressing the ER stress indicator to show that it can be used to monitor physiological and pathological ER stress in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Mori, K. Tripartite management of unfolded proteins in the endoplasmic reticulum. Cell 101, 451–454 (2000).
Ye, J. et al. ER stress induces cleavage of membrane-bound ATF6 by the same proteases that process SREBPs. Mol. Cell 6, 1355–1364 (2000).
Harding, H.P. et al. Regulated translation initiation controls stress-induced gene expression in mammalian cells. Mol. Cell 6, 1099–1108 (2000).
Tirasophon, W., Welihinda, A.A. & Kaufman, R.J. A stress response pathway from the endoplasmic reticulum to the nucleus requires a novel bifunctional protein kinase/endoribonuclease (Ire1p) in mammalian cells. Genes Dev. 12, 1812–1824 (1998).
Wang, X.Z. et al. Cloning of mammalian Ire1 reveals diversity in the ER stress responses. EMBO J. 17, 5708–5717 (1998).
Tirasophon, W. et al. The endoribonuclease activity of mammalian IRE-1 autoregulates its mRNA and is required for the unfolded protein response. Genes Dev. 14, 2725–2736 (2000).
Iwawaki, T et al. Translational control by the ER transmembrane kinase/ribonuclease IRE-1 under ER stress. Nat. Cell Biol. 3, 158–164 (2001).
Yoshida, H. et al. XBP-1 mRNA is induced by ATF6 and spliced by IRE-1 in response to ER stress to produce a highly active transcription factor. Cell 107, 881–891 (2001).
Shen, X. et al. Complementary signaling pathways regulate the unfolded protein response and are required for C. elegans development. Cell 107, 893–903 (2001).
Calfon, M. et al. IRE-1 couples endoplasmic reticulum load to secretory capacity by processing the XBP-1 mRNA. Nature 415, 92–96 (2002).
Urano, F. et al. Coupling of stress in the ER to activation of JNK protein kinases by transmembrane protein kinase IRE-1. Science 287, 664–666 (2000).
Nakagawa, T. et al. Caspase-12 mediates endoplasmic-reticulum-specific apoptosis and cytotoxicity by amyloid-β. Nature 403, 98–103 (2000).
Rao, R.V. et al. Coupling endoplasmic reticulum stress to the cell death program. J. Biol. Chem. 276, 33869–33874 (2001).
Outinen, P.A. et al. Characterization of the stress-inducing effects of homocysteine. Biochem. J. 332, 213–221 (1998).
Outinen, P.A. et al. Homocysteine-induced endoplasmic reticulum stress and growth arrest leads to specific changes in gene expression in human vascular endothelial cells. Blood 94, 959–967 (1999).
Duell, P.B. & Malinow, M.R. Homocysteine: an important risk factor for atherosclerotic vascular disease. Curr. Opin. Lipidol. 8, 28–34 (1997).
Mudd, S.H., Levy, H.L. & Kraus, P.J. Disorder of transsulfuration. in The Metabolic and Molecular Basis of Inherited Disease 8th edn. (eds. Scriver, C.R., Beaudet, A.L., Sly, W.S. & Valle, D.) 2007–2056 (McGraw-Hill, New York, 2001).
Refsum, H., Ueland, P.M., Nygrad, O. & Vollset, S.E. Homocysteine and cardiovascular disease. Annu. Rev. Med. 49, 31–62 (1998).
Paschen, W. & Frandsen, A. Endoplasmic reticulum dysfunction — a common denominator for cell injury in acute and degenerative diseases of the brain? J. Neurochem. 79, 719–725 (2001).
Katayama, T. et al. Presenilin-1 mutations downregulate the signaling pathway of the unfolded-protein response. Nat. Cell Biol. 1, 479–485 (1999).
Imai, Y. et al. An unfolded putative transmembrane polypeptide, which can lead to endoplasmic reticulum stress, is a substrate of Parkin. Cell 105, 891–902 (2001).
Nishitoh, H. et al. ASK1 is essential for endoplasmic reticulum stress-induced neuronal cell death triggered by expanded polyglutamine repeats. Genes Dev. 16, 1345–1355 (2002).
Reimold, A.M. et al. An essential role in liver development for transcription factor XBP-1. Genes Dev. 14, 152–157 (2000).
Nagai, T. et al. A variant of yellow fluorescent protein with fast and efficient maturation for cell-biological applications. Nat. Biotechnol. 20, 87–90 (2002).
Lee, A.H., Iwakoshi, N.N., Anderson, K.C. & Glimcher, L.H. Proteasome inhibitors disrupt the unfolded protein response in myeloma cells. Proc. Natl. Acad. Sci. USA 100, 9946–9951 (2003).
Zinszner, H. et al. CHOP is implicated in programmed cell death in response to impaired function of the endoplasmic reticulum. Genes Dev. 12, 982–995 (1998).
Acknowledgements
We thank A. Miyawaki for the venus cDNA and R. Kaufman for the human IRE-1α cDNA. T.I. was a research fellow of the Special Postdoctoral Researchers Program, RIKEN. This work was supported in part by grants from the Japanese Ministry of Education, Science, Sports, Culture, and Technology and by grants from the RIKEN Bioarchitect Research Project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Iwawaki, T., Akai, R., Kohno, K. et al. A transgenic mouse model for monitoring endoplasmic reticulum stress. Nat Med 10, 98–102 (2004). https://doi.org/10.1038/nm970
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm970
This article is cited by
-
Discovery of toxoflavin, a potent IRE1α inhibitor acting through structure-dependent oxidative inhibition
Acta Pharmacologica Sinica (2023)
-
P2X7 receptor activation mediates superoxide dismutase 1 (SOD1) release from murine NSC-34 motor neurons
Purinergic Signalling (2022)
-
Endoplasmic reticulum stress signals in the tumour and its microenvironment
Nature Reviews Cancer (2021)
-
The RNA landscape of the human placenta in health and disease
Nature Communications (2021)
-
Periplocin and cardiac glycosides suppress the unfolded protein response
Scientific Reports (2021)