Abstract
The transforming growth factor-β (TGF-β) signaling pathway serves critical functions in CNS development, but, apart from its proposed neuroprotective actions, its physiological role in the adult brain is unclear. We observed a prominent activation of TGF-β signaling in the adult dentate gyrus and expression of downstream Smad proteins in this neurogenic zone. Consistent with a function of TGF-β signaling in adult neurogenesis, genetic deletion of the TGF-β receptor ALK5 reduced the number, migration and dendritic arborization of newborn neurons. Conversely, constitutive activation of neuronal ALK5 in forebrain caused a marked increase in these aspects of neurogenesis and was associated with higher expression of c-Fos in newborn neurons and with stronger memory function. Our findings describe an unexpected role for ALK5-dependent TGF-β signaling as a regulator of the late stages of adult hippocampal neurogenesis, which may have implications for changes in neurogenesis during aging and disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Accession codes
References
Krieglstein, K., Zheng, F., Unsicker, K. & Alzheimer, C. More than being protective: functional roles for TGF-beta/activin signaling pathways at central synapses. Trends Neurosci. 34, 421–429 (2011).
Blobe, G.C., Schiemann, W.P. & Lodish, H.F. Role of transforming growth factor beta in human disease. N. Engl. J. Med. 342, 1350–1358 (2000).
Unsicker, K., Flanders, K.C., Cissel, D.S., Lafyatis, R. & Sporn, M.B. Transforming growth factor beta isoforms in the adult rat central and peripheral nervous system. Neuroscience 44, 613–625 (1991).
Shi, Y. & Massagué, J. Mechanisms of TGF-beta signaling from cell membrane to the nucleus. Cell 113, 685–700 (2003).
Feng, X.H. & Derynck, R. Specificity and versatility in TGF-β signaling through Smads. Annu. Rev. Cell Dev. Biol. 21, 659–693 (2005).
Tomoda, T. et al. Transforming growth factor-beta is a survival factor for neonate cortical neurons: coincident expression of type I receptors in developing cerebral cortices. Dev. Biol. 179, 79–90 (1996).
Falk, S. et al. Brain area–specific effect of TGF-beta signaling on Wnt-dependent neural stem cell expansion. Cell Stem Cell 2, 472–483 (2008).
Ishihara, A., Saito, H. & Abe, K. Transforming growth factor-beta 1 and -beta 2 promote neurite sprouting and elongation of cultured rat hippocampal neurons. Brain Res. 639, 21–25 (1994).
Ng, J. TGF-beta signals regulate axonal development through distinct Smad-independent mechanisms. Development 135, 4025–4035 (2008).
Yi, J.J., Barnes, A.P., Hand, R., Polleux, F. & Ehlers, M.D. TGF-beta signaling specifies axons during brain development. Cell 142, 144–157 (2010).
Unsicker, K. & Krieglstein, K. TGF-betas and their roles in the regulation of neuron survival. Adv. Exp. Med. Biol. 513, 353–374 (2002).
Tesseur, I. & Wyss-Coray, T. A role for TGF-beta signaling in neurodegeneration: evidence from genetically engineered models. Curr. Alzheimer Res. 3, 505–513 (2006).
Brionne, T.C., Tesseur, I., Masliah, E. & Wyss-Coray, T. Loss of TGF-beta1 leads to increased neuronal cell death and microgliosis in mouse brain. Neuron 40, 1133–1145 (2003).
Tesseur, I. et al. Deficiency in neuronal TGF-beta signaling promotes neurodegeneration and Alzheimer's pathology. J. Clin. Invest. 116, 3060–3069 (2006).
Buckwalter, M.S. et al. Chronically increased transforming growth factor-beta1 strongly inhibits hippocampal neurogenesis in aged mice. Am. J. Pathol. 169, 154–164 (2006).
Wachs, F.P. et al. Transforming growth factor-beta1 is a negative modulator of adult neurogenesis. J. Neuropathol. Exp. Neurol. 65, 358–370 (2006).
Deng, W., Aimone, J.B. & Gage, F.H. New neurons and new memories: how does adult hippocampal neurogenesis affect learning and memory? Nat. Rev. Neurosci. 11, 339–350 (2010).
Faigle, R. & Song, H. Signaling mechanisms regulating adult neural stem cells and neurogenesis. Biochim. Biophys. Acta 1830, 2435–2448 (2013).
Parent, J.M. Injury-induced neurogenesis in the adult mammalian brain. Neuroscientist 9, 261–272 (2003).
Duan, X., Kang, E., Liu, C.Y., Ming, G.L. & Song, H. Development of neural stem cell in the adult brain. Curr. Opin. Neurobiol. 18, 108–115 (2008).
Luo, J., Lin, A.H., Masliah, E. & Wyss-Coray, T. Bioluminescence imaging of Smad signaling in living mice shows correlation with excitotoxic neurodegeneration. Proc. Natl. Acad. Sci. USA 103, 18326–18331 (2006).
Lin, A.H. et al. Global analysis of Smad2/3-dependent TGF-beta signaling in living mice reveals prominent tissue-specific responses to injury. J. Immunol. 175, 547–554 (2005).
Plümpe, T. et al. Variability of doublecortin-associated dendrite maturation in adult hippocampal neurogenesis is independent of the regulation of precursor cell proliferation. BMC Neurosci. 7, 77 (2006).
Mazerbourg, S. & Hsueh, A.J. Genomic analyses facilitate identification of receptors and signaling pathways for growth differentiation factor 9 and related orphan bone morphogenetic protein/growth differentiation factor ligands. Hum. Reprod. Update 12, 373–383 (2006).
Wu, H.H. et al. Autoregulation of neurogenesis by GDF11. Neuron 37, 197–207 (2003).
Muzumdar, M.D., Tasic, B., Miyamichi, K., Li, L. & Luo, L. A global double-fluorescent Cre reporter mouse. Genesis 45, 593–605 (2007).
Marxreiter, F. et al. Changes in adult olfactory bulb neurogenesis in mice expressing the A30P mutant form of alpha-synuclein. Eur. J. Neurosci. 29, 879–890 (2009).
Imielski, Y. et al. Regrowing the adult brain: NF-kappaB controls functional circuit formation and tissue homeostasis in the dentate gyrus. PLoS ONE 7, e30838 (2012).
Mayford, M. et al. Control of memory formation through regulated expression of a CaMKII transgene. Science 274, 1678–1683 (1996).
Jessberger, S., Romer, B., Babu, H. & Kempermann, G. Seizures induce proliferation and dispersion of doublecortin-positive hippocampal progenitor cells. Exp. Neurol. 196, 342–351 (2005).
Kempermann, G., Gast, D., Kronenberg, G., Yamaguchi, M. & Gage, F.H. Early determination and long-term persistence of adult-generated new neurons in the hippocampus of mice. Development 130, 391–399 (2003).
Stevens, B. et al. The classical complement cascade mediates CNS synapse elimination. Cell 131, 1164–1178 (2007).
Hevner, R.F. et al. Tbr1 regulates differentiation of the preplate and layer 6. Neuron 29, 353–366 (2001).
Bury, F.J. et al. Xenopus BTBD6 and its Drosophila homologue lute are required for neuronal development. Dev. Dyn. 237, 3352–3360 (2008).
Resende, R.R., da Costa, J.L., Kihara, A.H., Adhikari, A. & Lorencon, E. Intracellular Ca2+ regulation during neuronal differentiation of murine embryonal carcinoma and mesenchymal stem cells. Stem Cells Dev. 19, 379–394 (2010).
Hirokawa, N., Noda, Y., Tanaka, Y. & Niwa, S. Kinesin superfamily motor proteins and intracellular transport. Nat. Rev. Mol. Cell Biol. 10, 682–696 (2009).
Guzowski, J.F. et al. Mapping behaviorally relevant neural circuits with immediate-early gene expression. Curr. Opin. Neurobiol. 15, 599–606 (2005).
Park, S.M., Jung, J.S., Jang, M.S., Kang, K.S. & Kang, S.K. Transforming growth factor-beta1 regulates the fate of cultured spinal cord–derived neural progenitor cells. Cell Prolif. 41, 248–264 (2008).
Prehn, J.H. et al. Regulation of neuronal Bcl2 protein expression and calcium homeostasis by transforming growth factor type beta confers wide-ranging protection on rat hippocampal neurons. Proc. Natl. Acad. Sci. USA 91, 12599–12603 (1994).
Krieglstein, K., Suter-Crazzolara, C., Fischer, W.H. & Unsicker, K. TGF-beta superfamily members promote survival of midbrain dopaminergic neurons and protect them against MPP+ toxicity. EMBO J. 14, 736–742 (1995).
Gleeson, J.G., Lin, P.T., Flanagan, L.A. & Walsh, C.A. Doublecortin is a microtubule-associated protein and is expressed widely by migrating neurons. Neuron 23, 257–271 (1999).
Gdalyahu, A. et al. DCX, a new mediator of the JNK pathway. EMBO J. 23, 823–832 (2004).
Feng, Y. & Walsh, C.A. Protein-protein interactions, cytoskeletal regulation and neuronal migration. Nat. Rev. Neurosci. 2, 408–416 (2001).
Kvajo, M. et al. A mutation in mouse Disc1 that models a schizophrenia risk allele leads to specific alterations in neuronal architecture and cognition. Proc. Natl. Acad. Sci. USA 105, 7076–7081 (2008).
Enomoto, A. et al. Roles of disrupted-in-schizophrenia 1–interacting protein girdin in postnatal development of the dentate gyrus. Neuron 63, 774–787 (2009).
Duan, X. et al. Disrupted-In-Schizophrenia 1 regulates integration of newly generated neurons in the adult brain. Cell 130, 1146–1158 (2007).
Porteous, D.J., Thomson, P., Brandon, N.J. & Millar, J.K. The genetics and biology of DISC1–an emerging role in psychosis and cognition. Biol. Psychiatry 60, 123–131 (2006).
Benes, F.M. et al. Regulation of the GABA cell phenotype in hippocampus of schizophrenics and bipolars. Proc. Natl. Acad. Sci. USA 104, 10164–10169 (2007).
Marín-Burgin, A. & Schinder, A.F. Requirement of adult-born neurons for hippocampus-dependent learning. Behav. Brain Res. 227, 391–399 (2012).
Larsson, J. et al. Abnormal angiogenesis but intact hematopoietic potential in TGF-beta type I receptor–deficient mice. EMBO J. 20, 1663–1673 (2001).
Wieser, R., Wrana, J.L. & Massague, J. GS domain mutations that constitutively activate T beta R-I, the downstream signaling component in the TGF-beta receptor complex. EMBO J. 14, 2199–2208 (1995).
Gossen, M. & Bujard, H. Tight control of gene expression in mammalian cells by tetracycline-responsive promoters. Proc. Natl. Acad. Sci. USA 89, 5547–5551 (1992).
Dittgen, T. et al. Lentivirus-based genetic manipulations of cortical neurons and their optical and electrophysiological monitoring in vivo. Proc. Natl. Acad. Sci. USA 101, 18206–18211 (2004).
Mosher, K.I. et al. Neural progenitor cells regulate microglia functions and activity. Nat. Neurosci. 15, 1485–1487 (2012).
Hughes, R.N. The value of spontaneous alternation behavior (SAB) as a test of retention in pharmacological investigations of memory. Neurosci. Biobehav. Rev. 28, 497–505 (2004).
Villeda, S.A. et al. The ageing systemic milieu negatively regulates neurogenesis and cognitive function. Nature 477, 90–94 (2011).
March, A., Borchelt, D., Golde, T. & Janus, C. Differences in memory development among C57BL/6NCrl, 129S2/SvPasCrl, and FVB/NCrl mice after delay and trace fear conditioning. Comp. Med. 64, 4–12 (2014).
Owen, E.H., Logue, S.F., Rasmussen, D.L. & Wehner, J.M. Assessment of learning by the Morris water task and fear conditioning in inbred mouse strains and F1 hybrids: implications of genetic background for single gene mutations and quantitative trait loci analyses. Neuroscience 80, 1087–1099 (1997).
Farley, S.J., McKay, B.M., Disterhoft, J.F. & Weiss, C. Reevaluating hippocampus-dependent learning in FVB/N mice. Behav. Neurosci. 125, 871–878 (2011).
Acknowledgements
We thank our laboratory members for discussions and comments, K. Lucin for editorial assistance, J. Luo for comments on data analysis, G. Bieri for providing brain sections of mT/mG mice crossed with Camk2a-cre mice, L. Boerma for helping mouse behavioral tests, P. Osten for providing pFCK(1.3)GW plasmid, and M. Lochrie and the Stanford Neuroscience Gene Vector and Virus Core for generating the viruses (supported by National Institute of Neurological Disorders and Stroke P30 core grant, NS069375-01A1). This work was supported by US National Institutes of Health grant AG27505 (T.W.-C.) and the Department of Veterans Affairs (T.W.-C.).
Author information
Authors and Affiliations
Contributions
Y.H. contributed to all aspects of the experiments and data analysis. H.Z. generated the ALK5CA mice. A.Y. assisted with the Alk5 shRNA experiments and in vivo data analysis. P.A.J. assisted with microarray data analysis. S.A.V. assisted with contextual fear conditioning experiments. O.O. assisted with doxycycline experiments. N.F. assisted with Y maze experiments. Y.H. and T.W.-C. wrote the manuscript. T.W.-C. supervised the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
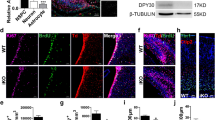
Supplementary Figure 1 Construction and characterization of ALK5cKO transgenic mice.
(a) Diagram of gene constructs used to create ALK5cKO mice. (b,c) Immunoblot of hippocampal lysates from 3-month-old ALK5loxP/loxP and ALK5cKOmice probed with antibodies against ALK5, NSE, and actin in (b) and quantification of ALK5 signal intensity normalized against NSE in (c), n = 2 mice per group, P = 0.0276. Full-length blots are presented in Supplementary Figure 10. The experiments were independently performed twice. (d,e) Representative tiled confocal images (d) and quantification (e) of the dentate gyrus from 3-month-old ALK5loxP/loxP (n = 8) and ALK5cKO(n = 7) mice immunostained for p-Smad2. P-Smad2 signals were normalized to background in (e). Three sections per mouse, P = 0.0433. (f) Representative orthogonal projections from confocal Z-stack images of the dentate gyrus from 3-month-old mT/mG mice before and after crossing with CamK2a-cre mice immunostained for eGFP in combination with Sox2 plus GFAP, MCM2, Tbr2, DCX plus PCNA, and NeuN. DAPI stains nuclei. Scale bar is 100 μm in (d) and 10 μm in (f). (g) Quantification of eGFP positive cells in different cell types detected in (f) from the dentate gyrus. (h) Quantification of eGFP positive and negative cells in individual cell types detected in (f) from the dentate gyrus. n = 4 mice, 3 sections per mouse in (g,h). Data are presented as mean ± s.d. in (c), mean ± s.e.m. in (e,g,h). *P < 0.05, ****P < 0.0001 Student's t-test in (c,e); one-way ANOVA, Dunnett's post-hoc test in (g).
Supplementary Figure 2 Deficiency of ALK5 is not affecting the neural stem cell pool and production of committed neurons.
(a,b) Immunohistochemical detection (a) and quantification (b) of Sox2 labeled cells in the dentate gyrus of 3-month-old ALK5loxP/loxP and ALK5cKO mice (n = 7 mice per group, 5 sections per mouse, P = 0.9023). (c) Quantification of the percentage of BrdU and DCX double-labeled cells in the dentate gyrus from 7-month-old ALK5loxP/loxP and ALK5cKO mice after short-term BrdU labeling (n = 5 mice per group, 5 sections per mouse, P = 0.8043). Experimental design inserted on top. (d) Quantification of total PCNA+ proliferating cells in the dentate gyrus of 3-month-old ALK5loxP/loxP and ALK5cKO mice (n = 7 mice per group, 5 sections per mouse, P = 0.8384) (e) Quantification of the percentage of DCX and PCNA double-labeled cells in the dentate gyrus of 3-month-old ALK5loxP/loxP and ALK5cKO mice (n = 7 mice per group, 5 sections per mouse, P = 0.3450). Scale bar is 100 μm. Data are presented as mean ± s.e.m. Student's t-test.
Supplementary Figure 3 Construction and characterization of cre lentivirus that decreases the survival of infected newborn neurons in ALK5loxP/loxP mouse dentate gyrus.
(a) Design of the lentiviral vectors. (b) Immunohistochemical detection of eGFP labeled lentivirus-infected cells in the dentate gyrus of stereotaxically injected 2-month-old ALK5loxP/loxP mice. (c,d) Representative confocal images (c) and quantification (d) of lentivirus-injected dentate gyrus from 3-month-old ALK5loxP/loxP mice immunostained for p-Smad2 and eGFP. P-Smad2 signals were normalized to signals in uninfected cells in (d). Green or blue dashed circles highlight the cell bodies of eGFP+ or eGFP– neurons. n = 27, 21, 24, 23 neurons from three mice total for eGFP–/LV-eGFP, eGFP+/LV-eGFP, eGFP–/LV-cre-eGFP, eGFP+/LV-cre-eGFP group, respectively. (e) Representative orthogonal projections from confocal Z-stack images of LV-cre-eGFP lentivirus-injected dentate gyrus of2-month-old ALK5loxP/loxP mice immunostained for eGFP in combination with Sox2 plus GFAP, MCM2, Tbr2, DCX plus PCNA, and NeuN. DAPI stains nuclei. Scale bar is 100 μm in (b) and 10 μm in (c,e). (f) Quantification of the number of lentivirus-infected eGFP and DCX double-labeled newborn neurons in the dentate gyrus from LV-cre-eGFP and LV-eGFP stereotaxically injected ALK5loxP/loxP mice with 2 months of age at 6, 14, and 21 dpi. Cell numbers were normalized to values at 6 dpi. n = 4 mice per group, 4 sections per mouse. *P = 0.0355. Data are presented as mean + s.d. in (d) and mean + s.e.m. in (f). ****P < 0.0001. One-way ANOVA, Tukey's post-hoc test (d); one-tailed Student's t-test (f).
Supplementary Figure 4 Construction and characterization of ALK5 shRNA retroviruses that decrease the survival of infected newborn neurons in C57BL/6 mouse dentate gyrus.
(a) Design of the retroviral vectors. MCS, multiple cloning site. (b) Immunoblot of lysates from HEK293T cells co-transfected with an expression vector for ALK5 and different shRNAs probed with antibodies against ALK5 and actin. Full-length blots are presented in Supplementary Figure 10. (c,d) Representative confocal images of retrovirus-injected dentate gyrus of 2-month-old C57BL/6 mice immunostained for eGFP and ALK5 at 7 dpi in (c) or eGFP and p-Smad2 at both 7 dpi and 14 dpi in (d). DAPI stains nuclei. Green or blue dashed circles highlight the cell bodies of eGFP+ or eGFP– neurons. Scale bar is 10 μm. (e) Quantification of p-Smad2 signal intensity from (d). P-Smad2 signals were normalized to signals in uninfected cells in (e). n = 20, 20, 23, 23 neurons from four mice total for eGFP–/shRNA-C, eGFP+/shRNA-C, eGFP–/shRNA-A1, eGFP+/shRNA-A1 group at 7 dpi, respectively. n = 15, 15, 14, 14 neurons from four mice total for eGFP–/shRNA-C, eGFP+/shRNA-C, eGFP–/shRNA-A1, eGFP+/shRNA-A1 group at 14 dpi, respectively. (f) Quantification of the number of virus-infected mCherry only and eGFP-labeled (eGFP only and eGFP/mCherry double-labeled) newborn neurons in the dentate gyrus from stereotaxically injected 2-month-old female C57BL/6 mice at 7 (n = 5 mice), 14 (n = 4 mice, P = 0.0485), and 21 dpi (n = 5 mice, P = 0.0021). Four sections per mouse. Cell numbers were normalized to values at 7 dpi. Data are presented as mean + s.d. in (e) and mean + s.e.m. in (f). *P < 0.05, **P < 0.01, ***P < 0.001. One-way ANOVA, Tukey's post-hoc test (e); Student's t-test (f).
Supplementary Figure 5 Knockdown of ALK5 by shRNA-A2 retrovirus in progenitor cells decelerates dendrite development of newborn neurons in the dentate gyrus.
(a,b) Brightfield immunohistochemical detection of virus-infected eGFP-immunostained cells (left) and confocal images of eGFP, DCX, and DAPI triple-labeled newborn neurons (right) in the dentate gyrus of stereotaxically injected 2-month-old C57BL/6 mice at 7 (a) and 14 dpi (b). Scale bar is 50 μm in brightfield immunohistochemical images and 20 μm in confocal images. (c) Quantification of dendritic length of virus-infected newborn neurons in the dentate gyrus at 7 dpi. (d,e) Quantification of dendritic length (d) and number (e) of virus-infected newborn neurons in the dentate gyrus at 14 dpi. n = 17, 12 neurons from two mice total for each group at 7 dpi. n = 13, 9 neurons from two mice total for each group at 14 dpi. Data are presented as mean + s.d. **P < 0.01, ***P < 0.001, Student's t-test.
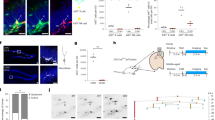
Supplementary Figure 6 Construction and characterization of ALK5CA transgenic mice.
(a) Diagram of gene constructs used to create ALK5CA mice. (b) Immunoblots and quantification of p-Smad2 in hippocampal lysates from 3-month-old ALK5Ctrl and ALK5CAmice probed with antibodies against p-Smad2, Smad2, HA, eGFP, and actin. P-Smad2 signal intensity was normalized against Smad2. n = 2 mice per group. Full-length blots are presented in Supplementary Figure 10. Data are presented as mean + s.d. **P = 0.0085, Student's t-test. The experiments were independently performed twice. (c) Representative orthogonal projections from confocal Z-stack images of the dentate gyrus of 4-month-old ALK5CA mice before and after treatment with doxycycline immunostained for eGFP and HA. (d) eGFP expression in the dentate gyrus (dotted line) of ALK5CA mice, but not of ALK5Ctrl mice detected by immunohistochemistry. (e) Representative orthogonal projections from confocal Z-stack images of the dentate gyrus of 3-month-old ALK5CAmice immunostained for eGFP, HA, and DCX. (f) Representative orthogonal projections from confocal Z-stack images of the dentate gyrus of 3-month-old ALK5CAmice immunostained for eGFP in combination with Sox2 plus GFAP, MCM2, Tbr2, DCX plus PCNA, and NeuN. (g) Scheme depicting expression patterns of p-Smad2 and CamK2a-driven genes during neuronal development based on characterization with specific neuronal development markers. DAPI stains nuclei. Scale bar is 100 μm in (d) and 10 μm in (c,e,f).
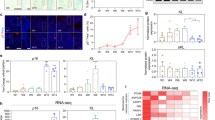
Supplementary Figure 7 Constitutive activation of ALK5 in forebrain neurons is not affecting neural stem cell pool and production of committed neurons, but is reducing apoptotic markers and increasing newborn neuron survival in the dentate gyrus.
(a) Quantification of Sox2+ cells in the SGZ of the dentate gyrus of 3-month-old ALK5Ctrl and ALK5CA mice (n = 5 mice per group, 5 sections per mouse, P = 0.2514). (b) Experimental design for long-term and short-term BrdU-labeling paradigms. (c) Quantification of the total number of BrdU labeled cells and the percentage of BrdU and DCX double-labeled cells in the dentate gyrus of ALK5Ctrl (n = 7) and ALK5CA (n = 6) mice (3-month-old) 1 day after BrdU administration. Five sections per mouse. (d) A representative confocal image of DAPI staining in the dentate gyrus of 3-month-old ALK5CA mice. Arrow points to an apoptotic cell with a pyknotic nucleus (condensed DAPI staining). Scale bar, 20 μm. (e) Immunoblots of hippocampal lysates from 3-month-old ALK5Ctrl (n = 5) and ALK5CA (n = 4) mice probed with antibodies against Bcl-2, Bax, and NSE. Full-length blots are presented in Supplementary Figure 10. (f) Quantification of BrdU and NeuN double-labeled newborn neurons in the dentate gyrus of 4-month-old ALK5Ctrl and ALK5CA mice 28 days after BrdU administration before and after doxycycline treatment (n = 4 mice per group, 5 sections per mouse). Doxycycline × genotype interaction F(1,12) = 2.783, P = 0.1211; doxycycline treatment F(1,12) = 2.036, P = 0.1791; genotype F(1,12) = 2.134, P = 0.1697; Data are presented as mean ± s.e.m. Student's t-test in (a,c). Two-way ANOVA in (f).
Supplementary Figure 8 Doxycycline treatment suppresses accelerated migration and dendritic development of newborn neurons in ALK5CA mice.
(a) Immunohistochemical detection of DCX labeled neurons in the dentate gyrus (dotted line) from 4-month-old ALK5Ctrl and ALK5CA mice before and after doxycyclline treatment. (b) Relative distribution of DCX labeled cells in ALK5Ctrland ALK5CA mice before and after doxycycline treatment in the ML and three evenly separated layers of the dentate gyrus. (c,d) Dendritic development as quantified by dendritic number (c) and length (d) of DCX labeled cells from ALK5Ctrland ALK5CA mice before and after doxycycline treatment. Scale bar, 50 μm. n = 4 mice per group. In (c), doxycycline × genotype interaction F(1,12) = 59.12, P < 0.0001; doxycycline treatment F(1,12) = 42.25, P < 0.0001; genotype F(1,12) = 34.91, P < 0.0001. In (d), doxycycline × genotype interaction F(1,12) = 40.28, P < 0.0001; doxycycline treatment F(1,12) = 27.99, P = 0.0002; genotype F(1,12) = 26.15, P = 0.0003. Data are presented as mean + s.e.m. ****P < 0.0001, one-way ANOVA, Tukey's post-hoc test (b); two-way ANOVA, Sidak's post-hoc test (c,d).
Supplementary Figure 9 ALK5CA mice do not show any changes in total number of entries in the Y maze or the percentage of freezing under pre-tone and tone conditions in fear conditioning compared with genotype-matched control mice.
(a) Total number of entries in the Y maze was measured from 3-month-old male WT, tTA, ALK5Ctrl, and ALK5CA mice. n = 18 mice per group. (b) Fear conditioning assessed by the percentage of freezing was displayed by 3-month-old male WT, tTA, ALK5Ctrl, and ALK5CA mice before tone and when re-exposed 24 hours later to the tone. n = 11 mice per group. In (b), time × genotype interaction F(3,40) = 0.1782, P = 0.9106; time F(1,40) = 330.9, P < 0.0001; genotype F(3,40) = 0.1832, P = 0.9072. One-way ANOVA, Tukey's post-hoc test (a); Two-way repeated-measures ANOVA, Bonferroni's post-hoc test (b).
Supplementary Figure 10 Full-length blots presented in figures. The bands of interest are displayed within the dashed boxes.
(a) Full-length blots of Figure 6c. (b) Full-length blots of Supplementary Figure 1b. (c) Full-length blots of Supplementary Figure 4b. (d) Full-length blots of Supplementary Figure 6b. (e) Full-length blots of Supplementary Figure 7e.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–10 and Supplementary Table 1 (PDF 4797 kb)
Supplementary Methods Checklist
(PDF 434 kb)
Rights and permissions
About this article
Cite this article
He, Y., Zhang, H., Yung, A. et al. ALK5-dependent TGF-β signaling is a major determinant of late-stage adult neurogenesis. Nat Neurosci 17, 943–952 (2014). https://doi.org/10.1038/nn.3732
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3732
This article is cited by
-
Overexpressed ski efficiently promotes neurorestoration, increases neuronal regeneration, and reduces astrogliosis after traumatic brain injury
Gene Therapy (2023)
-
GDF11 expressed in the adult brain negatively regulates hippocampal neurogenesis
Molecular Brain (2021)
-
Role of Neuroinflammation in the Establishment of the Neurogenic Microenvironment in Brain Diseases
Current Tissue Microenvironment Reports (2021)
-
Impact of Melatonin on Full-Term Fetal Brain Development and Transforming Growth Factor-β Level in a Rat Model of Preeclampsia
Reproductive Sciences (2021)
-
An important role of PHRF1 in dendritic architecture and memory formation by modulating TGF-β signaling
Scientific Reports (2020)