Abstract
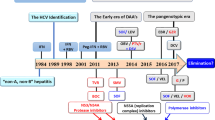
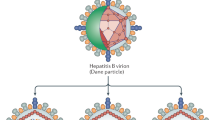
Immigration is fuelling a new reservoir of hepatitis D virus (HDV) in Europe, and hepatitis D still represents an important medical problem in the USA. The disease continues to be a major medical scourge in the developing world, in particular in countries such as Pakistan, Mongolia and Mauritania. New therapeutic strategies are being developed to disrupt interactions between HDV and its viral partner HBV, or with the host. Blocking or modifying the hepatitis B surface antigen (HBsAg) might interfere with the uptake or release of the hepatitis D virion; interference with host-mediated post-translational changes of proteins that are crucial to the HDV life cycle, such as prenylation, is another potential therapeutic option. At present, however, the only realistic option is to optimize IFN-α therapy. As eradication of HBsAg is the ultimate end point of therapy, long-term interferon administration might be required, raising an issue of tolerance in patients. Treatment with IFN-λ is a potential alternative approach to IFN-α; treatment of hepatitis C with this cytokine seems to cause fewer adverse effects than IFN-α and, therefore, might be more suitable for long-term treatment of HDV.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Rizzetto, M. Hepatitis D: thirty years after. J. Hepatol. 50, 1043–1050 (2009).
Taylor, J. M. Structure and replication of hepatitis delta virus RNA. Curr. Top. Microbiol. Immunol. 307, 1–23 (2006).
World Health Organization. Weekly epidemiological report. Hepatitis B position. 84, 405–420 (2009).
Rizzetto, M., Ponzetto, A. & Forzani, I. Epidemiology of hepatitis delta virus: overview. Prog. Clin. Biol. Res. 364, 1–20 (1991).
Rizzetto, M. & Ciancio, A. Epidemiology of hepatitis D. Semin. Liver Dis. 32, 211–219 (2012).
Huo, T. I. et al. Decreasing hepatitis D virus infection in Taiwan: an analysis of contributory factors. J. Gastroenterol. Hepatol. 12, 747–751 (1997).
Kao, J. H., Chen, P. J., Lai, M. Y. & Chen, D. S. Hepatitis D virus genotypes in intravenous drug users in Taiwan: decreasing prevalence and lack of correlation with hepatitis B virus genotypes. J. Clin. Microbiol. 40, 3047–3049 (2002).
Gaeta, G. B. et al. Hepatitis delta in Europe: vanishing or refreshing? Hepatology 46, 1312–1313 (2007).
Stroffolini, T. et al. Evolving clinical landscape of chronic hepatitis B: A multicenter Italian study. J. Med. Virol. 81, 1999–2006 (2009).
Heidrich, B. et al. Virological and clinical characteristics of delta hepatitis in Central Europe. J. Viral Hepat. 16, 883–894 (2009).
Cross, T. J. et al. The increasing prevalence of hepatitis delta virus (HDV) infection in South London. J. Med. Virol. 80, 277–282 (2008).
Le Gal, F. et al. Hepatitis D virus infection. Not a vanishing disease in Europe. Hepatology 45, 1332–1333 (2007).
Erhardt, A. et al. Socioepidemiological data on hepatitis delta in a German university clinic—increase in patients from Eastern Europe and the former Soviet Union. Z. Gastroenterol. 41, 523–526 (2003).
Bialek, S. R., Bower, W. A. & Mottram, K. Risk factors for hepatitis B in an outbreak of hepatitis B and D among injection drug users. J. Urban Health 82, 468–478 (2005).
Kucirka, L. M. et al. Prevalence, correlates, and viral dynamics of hepatitis delta among injection drug users. J. Infect. Dis. 202, 845–852 (2010).
Gish, R. G. et al. Coinfection with hepatitis B and D: epidemiology, prevalence and disease in patients in Northern California. J. Gastroenterol. Hepatol. http://dx.doi.org/10.1111/jgh.12217.
Holmberg, S. D. & Ward, J. W. Hepatitis delta: seek and ye shall find. J. Infect. Dis. 202, 822–824 (2010).
Niro, G. A., Rosina, F. & Rizzetto, M. Treatment of hepatitis D. J. Viral Hepat. 12, 2–9 (2005).
Farci, P. et al. Treatment of chronic hepatitis D with interferon α-2a. N. Engl. J. Med. 330, 88–94 (1994).
Ciancio, A. & Rizzetto, M. PEG-IFN for the treatment of hepatitis D. Nat. Rev. Gastroenterol. Hepatol. 8, 304–306 (2011).
Castelnau, C. et al. Efficacy of peginterferon α-2b in chronic hepatitis delta: relevance of quantitative RT-PCR for follow-up. Hepatology 44, 728–735 (2006).
Niro, G. A. et al. Pegylated interferon α-2b as monotherapy or in combination with ribavirin in chronic hepatitis delta. Hepatology 44, 713–720 (2006).
Erhardt, A. et al. Treatment of chronic hepatitis delta with pegylated interferon-α2b. Liver Int. 26, 805–810 (2006).
Wedemeyer, H. et al. Peginterferon plus adefovir versus either drug alone for hepatitis delta. N. Engl. J. Med. 364, 322–331 (2011).
Heidrich, B. et al. Long-term follow-up after peg-ifna2a based therapy of chronic hepatitis delta [abstract]. J. Hepatol. 58 (Suppl. 1), S20 (2013).
Niro, G. A. et al. Lamivudine therapy in chronic delta hepatitis: a multicentre randomized-controlled pilot study. Aliment. Pharmacol. Ther. 22, 227–232 (2005).
Yurdaydin, C. et al. Treatment of chronic delta hepatitis with lamivudine vs lamivudine + interferon vs interferon. J. Viral Hepat. 15, 314–321 (2008).
Kabaçam, G. et al. Entecavir treatment of chronic hepatitis D. Clin. Infect. Dis. 55, 645–650 (2012).
Kao, J. H. Diagnosis of hepatitis B virus infection through serological and virological markers. Expert Rev. Gastroenterol. Hepatol. 2, 553–562 (2008).
Lai, M. M. RNA replication without RNA-dependent RNA polymerase: surprises from hepatitis delta virus. J. Virol. 79, 7951–7958 (2005).
Singh, S. et al. Design of potential siRNA molecules for hepatitis delta virus gene silencing. Bioinformation 8, 749–757 (2012).
Chang, J. & Taylor, J. M. Susceptibility of human hepatitis delta virus RNAs to small interfering RNA action. J. Virol. 77, 9728–9731 (2003).
Sureau, C. The role of the HBV envelope proteins in the HDV replication cycle. Curr. Top. Microbiol. Immunol. 307, 113–131 (2006).
Abou-Jaoudé, G. & Sureau, C. Entry of hepatitis delta virus requires the conserved cysteine residues of the hepatitis B virus envelope protein antigenic loop and is blocked by inhibitors of thiol-disulfide exchange. J. Virol. 81, 13057–13066 (2007).
Lütgehetmann, M. et al. Humanized chimeric uPA mouse model for the study of hepatitis B and D virus interactions and preclinical drug evaluation. Hepatology 55, 685–694 (2012).
Komla-Soukha, I. & Sureau, C. A tryptophan-rich motif in the carboxyl terminus of the small envelope protein of hepatitis B virus is central to the assembly of hepatitis delta virus particles. J. Virol. 80, 4648–4655 (2006).
Vietheer, P. T., Netter, H. J., Sozzi, T. & Bartholomeusz, A. Failure of the lamivudine-resistant rtM204I hepatitis B virus mutants to efficiently support hepatitis delta virus secretion. J. Virol. 79, 6570–6573 (2005).
Andreone, P., Spertini, F. & Negro, F. Lamivudine-associated remission of chronic hepatitis delta. Ann. Intern. Med. 132, 598 (2000).
Shih, H. H. et al. Hepatitis B surface antigen levels and sequences of natural hepatitis B virus variants influence the assembly and secretion of hepatitis d virus. J. Virol. 82, 2250–2264 (2008).
Huang, W. H., Chen, C. W., Wu, H. L. & Chen, P. J. Post-translational modification of delta antigen of hepatitis D virus. Curr. Top. Microbiol. Immunol. 307, 91–112 (2006).
Glenn, J. S. Prenylation of HDAg and antiviral drug development. Curr. Top. Microbiol. Immunol. 307, 133–149 (2006).
Bordier, B. B. et al. A prenylation inhibitor prevents production of infectious hepatitis delta virus particles. J. Virol. 76, 10465–10472 (2002).
Bordier, B. B. et al. In vivo antiviral efficacy of prenylation inhibitors against hepatitis delta virus. J. Clin. Invest. 112, 407–414 (2003).
Ouzan, D., Pénaranda, G., Joly, H. & Halfon, P. Optimized HBsAg titer monitoring improves interferon therapy in patients with chronic delta. J. Hepatol. 58, 1258–1259 (2013).
Muir, A. J. et al. Peginterferon λ-1a (λ) compared with peginterferon α-2a (α) in treatment-naive patients with HCV genotypes 1 or 4: SVR24 results from EMERGE phase 2b [abstract]. J. Hepatology 56, 299A (2012).
Author information
Authors and Affiliations
Contributions
Both authors contributed equally to all aspects of the generation of this article.
Corresponding author
Ethics declarations
Competing interests
M. Rizzetto is on an Advisory Board for Bristol–Myers Squibb, Janssen and MSD. A. Ciancio declares no competing interests.
PowerPoint slides
Rights and permissions
About this article
Cite this article
Ciancio, A., Rizzetto, M. Chronic hepatitis D at a standstill: where do we go from here?. Nat Rev Gastroenterol Hepatol 11, 68–71 (2014). https://doi.org/10.1038/nrgastro.2013.164
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrgastro.2013.164
This article is cited by
-
Development of quantitative multiplex RT-qPCR one step assay for detection of hepatitis delta virus
Scientific Reports (2023)
-
Enveloped viruses distinct from HBV induce dissemination of hepatitis D virus in vivo
Nature Communications (2019)
-
High prevalence of hepatitis delta virus in Cameroon
Scientific Reports (2018)
-
Hepatitis delta: virological and clinical aspects
Virology Journal (2017)
-
A novel toolbox for the in vitro assay of hepatitis D virus infection
Scientific Reports (2017)