Abstract
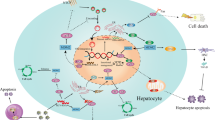
The development of hepatocellular carcinoma (HCC) in persons who are persistently infected with hepatitis C virus (HCV) is a growing problem worldwide. Current antiviral therapies are not effective in many patients with chronic hepatitis C, and a greater understanding of the factors leading to progression of HCC will be necessary to design novel approaches to prevention of HCV-associated HCC. The lack of a small animal model of chronic HCV infection has hampered understanding of these factors. As HCV is an RNA virus with little potential for integration of its genetic material into the host genome, the mechanisms underlying HCV promotion of cancer are likely to differ from other models of viral carcinogenesis. In patients persistently infected with HCV, chronic inflammation resulting from immune responses against infected hepatocytes is associated with progressive fibrosis and cirrhosis. Cirrhosis is an important risk factor for HCC independent of HCV infection, and a majority of HCV-associated HCC arises in the setting of cirrhosis. However, a significant minority arises in the absence of cirrhosis, indicating that cirrhosis is not a prerequisite for cancer. Other lines of evidence suggest that direct, virus-specific mechanisms may be involved. Transgenic mice expressing HCV proteins develop cancer in the absence of inflammation or immune recognition of the transgene. In vitro studies have revealed multiple interactions of HCV-encoded proteins with cell cycle regulators and tumor suppressor proteins, raising the possibility that HCV can disrupt control of cellular proliferation, or impair the cell's response to DNA damage. A combination of virus-specific, host genetic, environmental and immune-related factors are likely to determine the progression to HCC in patients who are chronically infected with HCV. Here, we summarize current knowledge of the virus-specific mechanisms that may contribute to HCV-associated HCC.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Centers for Disease Control and Prevention (CDC) (2010). Hepatocellular carcinoma-United States, 2001–2006. MMWR Morb Mortal Wkly Rep 59: 517–520.
Altekruse SF, McGlynn KA, Reichman ME . (2009). Hepatocellular carcinoma incidence, mortality, and survival trends in the United States from 1975 to 2005. J Clin Oncol 27: 1485–1491.
Angus AG, Dalrymple D, Boulant S, McGivern DR, Clayton RF, Scott MJ et al. (2010). Requirement of cellular DDX3 for hepatitis C virus replication is unrelated to its interaction with the viral core protein. J Gen Virol 91: 122–132.
Ariumi Y, Kuroki M, Abe K, Dansako H, Ikeda M, Wakita T et al. (2007). DDX3 DEAD-box RNA helicase is required for hepatitis C virus RNA replication. J Virol 81: 13922–13926.
Ariumi Y, Kuroki M, Dansako H, Abe K, Ikeda M, Wakita T et al. (2008). The DNA damage sensors ataxia-telangiectasia mutated kinase and checkpoint kinase 2 are required for hepatitis C virus RNA replication. J Virol 82: 9639–9646.
Armstrong GL, Wasley A, Simard EP, McQuillan GM, Kuhnert WL, Alter MJ . (2006). The prevalence of hepatitis C virus infection in the United States, 1999 through 2002. Ann Intern Med 144: 705–714.
Azechi H, Nishida N, Fukuda Y, Nishimura T, Minata M, Katsuma H et al. (2001). Disruption of the p16/cyclin D1/retinoblastoma protein pathway in the majority of human hepatocellular carcinomas. Oncology 60: 346–354.
Bai S, Nasser MW, Wang B, Hsu SH, Datta J, Kutay H et al. (2009). MicroRNA-122 inhibits tumorigenic properties of hepatocellular carcinoma cells and sensitizes these cells to sorafenib. J Biol Chem 284: 32015–32027.
Baroni GS, Pastorelli A, Manzin A, Benedetti A, Marucci L, Solforosi L et al. (1999). Hepatic stellate cell activation and liver fibrosis are associated with necroinflammatory injury and Th1-like response in chronic hepatitis C. Liver 19: 212–219.
Bartsch H, Nair J . (2004). Oxidative stress and lipid peroxidation-derived DNA-lesions in inflammation driven carcinogenesis. Cancer Detect Prev 28: 385–391.
Bataller R, Paik YH, Lindquist JN, Lemasters JJ, Brenner DA . (2004). Hepatitis C virus core and nonstructural proteins induce fibrogenic effects in hepatic stellate cells. Gastroenterology 126: 529–540.
Bates GJ, Nicol SM, Wilson BJ, Jacobs AM, Bourdon JC, Wardrop J et al. (2005). The DEAD box protein p68: a novel transcriptional coactivator of the p53 tumour suppressor. EMBO J 24: 543–553.
Battaglia S, Benzoubir N, Nobilet S, Charneau P, Samuel D, Zignego AL et al. (2009). Liver cancer-derived hepatitis C virus core proteins shift TGF-Beta responses from tumor suppression to epithelial-mesenchymal transition. PLoS ONE 4: e4355.
Bigger CB, Guerra KM, Brasky KM, Hubbard G, Beard MR, Luxon B et al. (2004). Intrahepatic gene expression during chronic hepatitis C virus infection in chimpanzees. J Virol 78: 13779–13792.
Botlagunta M, Vesuna F, Mironchik Y, Raman A, Lisok A, Winnard Jr P et al. (2008). Oncogenic role of DDX3 in breast cancer biogenesis. Oncogene 27: 3912–3922.
Bowen DG, Walker CM . (2005). Adaptive immune responses in acute and chronic hepatitis C virus infection. Nature 436: 946–952.
Bralet MP, Regimbeau JM, Pineau P, Dubois S, Loas G, Degos F et al. (2000). Hepatocellular carcinoma occurring in nonfibrotic liver: epidemiologic and histopathologic analysis of 80 French cases. Hepatology 32: 200–204.
Brenner DA . (2009). Molecular pathogenesis of liver fibrosis. Trans Am Clin Climatol Assoc 120: 361–368.
Bressac B, Galvin KM, Liang TJ, Isselbacher KJ, Wands JR, Ozturk M . (1990). Abnormal structure and expression of p53 gene in human hepatocellular carcinoma. Proc Natl Acad Sci USA 87: 1973–1977.
Chang J, Nicolas E, Marks D, Sander C, Lerro A, Buendia MA et al. (2004). miR-122, a mammalian liver-specific microRNA, is processed from hcr mRNA and may downregulate the high affinity cationic amino acid transporter CAT-1. RNA Biol 1: 106–113.
Chang PC, Chi CW, Chau GY, Li FY, Tsai YH, Wu JC et al. (2006). DDX3, a DEAD box RNA helicase, is deregulated in hepatitis virus-associated hepatocellular carcinoma and is involved in cell growth control. Oncogene 25: 1991–2003.
Chao CH, Chen CM, Cheng PL, Shih JW, Tsou AP, Lee YH . (2006). DDX3, a DEAD box RNA helicase with tumor growth-suppressive property and transcriptional regulation activity of the p21waf1/cip1 promoter, is a candidate tumor suppressor. Cancer Res 66: 6579–6588.
Chellappan SP, Hiebert S, Mudryj M, Horowitz JM, Nevins JR . (1991). The E2F transcription factor is a cellular target for the RB protein. Cell 65: 1053–1061.
Cheng PL, Chang MH, Chao CH, Lee YH . (2004). Hepatitis C viral proteins interact with Smad3 and differentially regulate TGF-beta/Smad3-mediated transcriptional activation. Oncogene 23: 7821–7838.
Chou AH, Tsai HF, Wu YY, Hu CY, Hwang LH, Hsu PI et al. (2005). Hepatitis C virus core protein modulates TRAIL-mediated apoptosis by enhancing Bid cleavage and activation of mitochondria apoptosis signaling pathway. J Immunol 174: 2160–2166.
Classon M, Harlow E . (2002). The retinoblastoma tumour suppressor in development and cancer. Nat Rev Cancer 2: 910–917.
Coulouarn C, Factor VM, Andersen JB, Durkin ME, Thorgeirsson SS . (2009). Loss of miR-122 expression in liver cancer correlates with suppression of the hepatic phenotype and gain of metastatic properties. Oncogene 28: 3526–3536.
Cross TJ, Rashid MM, Berry PA, Harrison PM . (2010). The importance of steatosis in chronic hepatitis C infection and its management: a review. Hepatol Res 40: 237–247.
Davis GL, Alter MJ, El-Serag H, Poynard T, Jennings LW . (2010). Aging of hepatitis C virus (HCV)-infected persons in the United States: a multiple cohort model of HCV prevalence and disease progression. Gastroenterology 138: 513–521, 521 e511-516.
DeCaprio JA, Ludlow JW, Figge J, Shew JY, Huang CM, Lee WH et al. (1988). SV40 large tumor antigen forms a specific complex with the product of the retinoblastoma susceptibility gene. Cell 54: 275–283.
Degos F, Christidis C, Ganne-Carrie N, Farmachidi JP, Degott C, Guettier C et al. (2000). Hepatitis C virus related cirrhosis: time to occurrence of hepatocellular carcinoma and death. Gut 47: 131–136.
Deng L, Adachi T, Kitayama K, Bungyoku Y, Kitazawa S, Ishido S et al. (2008). Hepatitis C virus infection induces apoptosis through a Bax-triggered, mitochondrion-mediated, caspase 3-dependent pathway. J Virol 82: 10375–10385.
Deng L, Nagano-Fujii M, Tanaka M, Nomura-Takigawa Y, Ikeda M, Kato N et al. (2006). NS3 protein of Hepatitis C virus associates with the tumour suppressor p53 and inhibits its function in an NS3 sequence-dependent manner. J Gen Virol 87: 1703–1713.
Dharel N, Kato N, Muroyama R, Taniguchi H, Otsuka M, Wang Y et al. (2008). Potential contribution of tumor suppressor p53 in the host defense against hepatitis C virus. Hepatology 47: 1136–1149.
Diamond DL, Jacobs JM, Paeper B, Proll SC, Gritsenko MA, Carithers Jr RL et al. (2007). Proteomic profiling of human liver biopsies: hepatitis C virus-induced fibrosis and mitochondrial dysfunction. Hepatology 46: 649–657.
Dolganiuc A, Oak S, Kodys K, Golenbock DT, Finberg RW, Kurt-Jones E et al. (2004). Hepatitis C core and nonstructural 3 proteins trigger toll-like receptor 2-mediated pathways and inflammatory activation. Gastroenterology 127: 1513–1524.
Dutta U, Kench J, Byth K, Khan MH, Lin R, Liddle C et al. (1998). Hepatocellular proliferation and development of hepatocellular carcinoma: a case-control study in chronic hepatitis C. Hum Pathol 29: 1279–1284.
Dyson N, Howley PM, Munger K, Harlow E . (1989). The human papilloma virus-16 E7 oncoprotein is able to bind to the retinoblastoma gene product. Science 243: 934–937.
Edamoto Y, Hara A, Biernat W, Terracciano L, Cathomas G, Riehle HM et al. (2003). Alterations of RB1, p53 and Wnt pathways in hepatocellular carcinomas associated with hepatitis C, hepatitis B and alcoholic liver cirrhosis. Int J Cancer 106: 334–341.
Elmen J, Lindow M, Schutz S, Lawrence M, Petri A, Obad S et al. (2008). LNA-mediated microRNA silencing in non-human primates. Nature 452: 896–899.
Erdtmann L, Franck N, Lerat H, Le Seyec J, Gilot D, Cannie I et al. (2003). The hepatitis C virus NS2 protein is an inhibitor of CIDE-B-induced apoptosis. J Biol Chem 278: 18256–18264.
Farinati F, Cardin R, D'Errico A, De Maria N, Naccarato R, Cecchetto A et al. (1996). Hepatocyte proliferative activity in chronic liver damage as assessed by the monoclonal antibody MIB1 Ki67 in archival material: the role of etiology, disease activity, iron, and lipid peroxidation. Hepatology 23: 1468–1475.
Fornari F, Gramantieri L, Giovannini C, Veronese A, Ferracin M, Sabbioni S et al. (2009). MiR-122/cyclin G1 interaction modulates p53 activity and affects doxorubicin sensitivity of human hepatocarcinoma cells. Cancer Res 69: 5761–5767.
Fox JG, Feng Y, Theve EJ, Raczynski AR, Fiala JL, Doernte AL et al. (2010). Gut microbes define liver cancer risk in mice exposed to chemical and viral transgenic hepatocarcinogens. Gut 59: 88–97.
Furutani T, Hino K, Okuda M, Gondo T, Nishina S, Kitase A et al. (2006). Hepatic iron overload induces hepatocellular carcinoma in transgenic mice expressing the hepatitis C virus polyprotein. Gastroenterology 130: 2087–2098.
Gale Jr M, Katze MG . (1998). Molecular mechanisms of interferon resistance mediated by viral-directed inhibition of PKR, the interferon-induced protein kinase. Pharmacol Ther 78: 29–46.
Gale Jr M, Kwieciszewski B, Dossett M, Nakao H, Katze MG . (1999). Antiapoptotic and oncogenic potentials of hepatitis C virus are linked to interferon resistance by viral repression of the PKR protein kinase. J Virol 73: 6506–6516.
Gale Jr MJ, Korth MJ, Tang NM, Tan SL, Hopkins DA, Dever TE et al. (1997). Evidence that hepatitis C virus resistance to interferon is mediated through repression of the PKR protein kinase by the nonstructural 5A protein. Virology 230: 217–227.
Gao Y, He Y, Ding J, Wu K, Hu B, Liu Y et al. (2009). An insertion/deletion polymorphism at miRNA-122-binding site in the interleukin-1alpha 3′ untranslated region confers risk for hepatocellular carcinoma. Carcinogenesis 30: 2064–2069.
Ghosh AK, Steele R, Meyer K, Ray R, Ray RB . (1999). Hepatitis C virus NS5A protein modulates cell cycle regulatory genes and promotes cell growth. J Gen Virol 80 (Part 5): 1179–1183.
Goh PY, Tan YJ, Lim SP, Tan YH, Lim SG, Fuller-Pace F et al. (2004). Cellular RNA helicase p68 relocalization and interaction with the hepatitis C virus (HCV) NS5B protein and the potential role of p68 in HCV RNA replication. J Virol 78: 5288–5298.
Gong G, Waris G, Tanveer R, Siddiqui A . (2001). Human hepatitis C virus NS5A protein alters intracellular calcium levels, induces oxidative stress, and activates STAT-3 and NF-kappa B. Proc Natl Acad Sci USA 98: 9599–9604.
Gramantieri L, Ferracin M, Fornari F, Veronese A, Sabbioni S, Liu CG et al. (2007). Cyclin G1 is a target of miR-122a, a microRNA frequently down-regulated in human hepatocellular carcinoma. Cancer Res 67: 6092–6099.
Henke JI, Goergen D, Zheng J, Song Y, Schuttler CG, Fehr C et al. (2008). microRNA-122 stimulates translation of hepatitis C virus RNA. EMBO J 27: 3300–3310.
Hernando E, Nahle Z, Juan G, Diaz-Rodriguez E, Alaminos M, Hemann M et al. (2004). Rb inactivation promotes genomic instability by uncoupling cell cycle progression from mitotic control. Nature 430: 797–802.
Honda A, Arai Y, Hirota N, Sato T, Ikegaki J, Koizumi T et al. (1999). Hepatitis C virus structural proteins induce liver cell injury in transgenic mice. J Med Virol 59: 281–289.
Honda M, Kaneko S, Shimazaki T, Matsushita E, Kobayashi K, Ping LH et al. (2000). Hepatitis C virus core protein induces apoptosis and impairs cell-cycle regulation in stably transformed Chinese hamster ovary cells. Hepatology 31: 1351–1359.
Ishido S, Hotta H . (1998). Complex formation of the nonstructural protein 3 of hepatitis C virus with the p53 tumor suppressor. FEBS Lett 438: 258–262.
Jangra RK, Yi M, Lemon SM . (2010a). DDX6 (Rck/p54) is required for efficient hepatitis C virus replication but not IRES-directed translation. J Virol 84: 6810–6824.
Jangra RK, Yi M, Lemon SM . (2010b). miR-122 regulation of hepatitis C virus translation and infectious virus production. J Virol 84: 6615–6625.
Jopling CL, Yi M, Lancaster AM, Lemon SM, Sarnow P . (2005). Modulation of hepatitis C virus RNA abundance by a liver-specific MicroRNA. Science 309: 1577–1581.
Jopling CL, Schutz S, Sarnow P . (2008). Position-dependent function for a tandem microRNA miR-122-binding site located in the hepatitis C virus RNA genome. Cell Host Microbe 4: 77–85.
Joyce MA, Walters KA, Lamb SE, Yeh MM, Zhu LF, Kneteman N et al. (2009). HCV induces oxidative and ER stress, and sensitizes infected cells to apoptosis in SCID/Alb-uPA mice. PLoS Pathogens 5: e1000291.
Kamegaya Y, Hiasa Y, Zukerberg L, Fowler N, Blackard JT, Lin W et al. (2005). Hepatitis C virus acts as a tumor accelerator by blocking apoptosis in a mouse model of hepatocarcinogenesis. Hepatology 41: 660–667.
Kao CF, Chen SY, Chen JY, Wu Lee YH . (2004). Modulation of p53 transcription regulatory activity and post-translational modification by hepatitis C virus core protein. Oncogene 23: 2472–2483.
Kato T, Miyamoto M, Date T, Yasui K, Taya C, Yonekawa H et al. (2003). Repeated hepatocyte injury promotes hepatic tumorigenesis in hepatitis C virus transgenic mice. Cancer Sci 94: 679–685.
Kawamura T, Furusaka A, Koziel MJ, Chung RT, Wang TC, Schmidt EV et al. (1997). Transgenic expression of hepatitis C virus structural proteins in the mouse. Hepatology 25: 1014–1021.
Keasler VV, Lerat H, Madden CR, Finegold MJ, McGarvey MJ, Mohammed EM et al. (2006). Increased liver pathology in hepatitis C virus transgenic mice expressing the hepatitis B virus X protein. Virology 347: 466–475.
Khidr L, Chen PL . (2006). RB, the conductor that orchestrates life, death and differentiation. Oncogene 25: 5210–5219.
Kim TY, Lee KH, Chang S, Chung C, Lee HW, Yim J et al. (2003). Oncogenic potential of a dominant negative mutant of interferon regulatory factor 3. J Biol Chem 278: 15272–15278.
Klopstock N, Katzenellenbogen M, Pappo O, Sklair-Levy M, Olam D, Mizrahi L et al. (2009). HCV tumor promoting effect is dependent on host genetic background. PloS One 4: e5025.
Koike K, Moriya K, Ishibashi K, Matsuura Y, Suzuki T, Saito I et al. (1995). Expression of hepatitis C virus envelope proteins in transgenic mice. J Gen Virol 76 (Part 12): 3031–3038.
Korenaga M, Wang T, Li Y, Showalter LA, Chan T, Sun J et al. (2005). Hepatitis C virus core protein inhibits mitochondrial electron transport and increases reactive oxygen species (ROS) production. J Biol Chem 280: 37481–37488.
Koskinas J, Petraki K, Kavantzas N, Rapti I, Kountouras D, Hadziyannis S . (2005). Hepatic expression of the proliferative marker Ki-67 and p53 protein in HBV or HCV cirrhosis in relation to dysplastic liver cell changes and hepatocellular carcinoma. J Viral Hepat 12: 635–641.
Krutzfeldt J, Rajewsky N, Braich R, Rajeev KG, Tuschl T, Manoharan M et al. (2005). Silencing of microRNAs in vivo with ′antagomirs′. Nature 438: 685–689.
Kutay H, Bai S, Datta J, Motiwala T, Pogribny I, Frankel W et al. (2006). Downregulation of miR-122 in the rodent and human hepatocellular carcinomas. J Cell Biochem 99: 671–678.
Kwun HJ, Jung EY, Ahn JY, Lee MN, Jang KL . (2001). p53-dependent transcriptional repression of p21(waf1) by hepatitis C virus NS3. J Gen Virol 82: 2235–2241.
Lai CK, Jeng KS, Machida K, Cheng YS, Lai MM . (2008). Hepatitis C virus NS3/4A protein interacts with ATM, impairs DNA repair and enhances sensitivity to ionizing radiation. Virology 370: 295–309.
Lan KH, Sheu ML, Hwang SJ, Yen SH, Chen SY, Wu JC et al. (2002). HCV NS5A interacts with p53 and inhibits p53-mediated apoptosis. Oncogene 21: 4801–4811.
Lanford RE, Hildebrandt-Eriksen ES, Petri A, Persson R, Lindow M, Munk ME et al. (2010). Therapeutic silencing of microRNA-122 in primates with chronic hepatitis C virus infection. Science 327: 198–201.
Lanford RE, Lemon SM, Walker C . (2011). The chimpanzee model of hepatitis C infections and small animal surrogates. In: He Y, Tan T (eds). Hepatitis C Antiviral Drug Discovery & Development. Horizons Scientific Press: Norwich. pp 99–132.
Lavanchy D . (2009). The global burden of hepatitis C. Liver Int 29 (Suppl 1): 74–81.
Lee SH, Kim YK, Kim CS, Seol SK, Kim J, Cho S et al. (2005). E2 of hepatitis C virus inhibits apoptosis. J Immunol 175: 8226–8235.
Lemon SM, Walker C, Alter MJ, Yi M . (2007). Hepatitis C viruses. In: Knipe D, Howley P, Griffin DE, Martin MA, Lamb RA, Roizman B et al (eds). Fields Virology, 5th edn. Lippincott, Williams & Wilkins: Philadelphia. pp 1253–1304.
Lemon SM . (2010). Induction and evasion of innate antiviral responses by hepatitis C virus. J Biol Chem 285: 22741–22747.
Lentini L, Pipitone L, Di LA . (2002). Functional inactivation of pRB results in aneuploid mammalian cells after release from a mitotic block. Neoplasia 4: 380–387.
Lerat H, Honda M, Beard MR, Loesch K, Sun J, Yang Y et al. (2002). Steatosis and liver cancer in transgenic mice expressing the structural and nonstructural proteins of hepatitis C virus. Gastroenterology 122: 352–365.
Li K, Foy E, Ferreon JC, Nakamura M, Ferreon AC, Ikeda M et al. (2005a). Immune evasion by hepatitis C virus NS3/4A protease-mediated cleavage of the Toll-like receptor 3 adaptor protein TRIF. Proc Natl Acad Sci USA 102: 2992–2997.
Li K, Prow T, Lemon SM, Beard MR . (2002). Cellular response to conditional expression of hepatitis C virus core protein in Huh7 cultured human hepatoma cells. Hepatology 35: 1237–1246.
Li XD, Sun L, Seth RB, Pineda G, Chen ZJ . (2005b). Hepatitis C virus protease NS3/4A cleaves mitochondrial antiviral signaling protein off the mitochondria to evade innate immunity. Proc Natl Acad Sci USA 102: 17717–17722.
Liang Y, Shilagard T, Xiao SY, Snyder N, Lau D, Cicalese L et al. (2009). Visualizing hepatitis C virus infections in human liver by two-photon microscopy. Gastroenterology 137: 1448–1458.
Lindenbach BD, Rice CM . (2005). Unravelling hepatitis C virus replication from genome to function. Nature 436: 933–938.
Liu X, Wang T, Wakita T, Yang W . (2010). Systematic identification of microRNA and messenger RNA profiles in hepatitis C virus-infected human hepatoma cells. Virology 398: 57–67.
Lohmann V, Korner F, Koch J, Herian U, Theilmann L, Bartenschlager R . (1999). Replication of subgenomic hepatitis C virus RNAs in a hepatoma cell line. Science 285: 110–113.
Lok AS, Seeff LB, Morgan TR, di Bisceglie AM, Sterling RK, Curto TM et al. (2009). Incidence of hepatocellular carcinoma and associated risk factors in hepatitis C-related advanced liver disease. Gastroenterology 136: 138–148.
Lu W, Lo SY, Chen M, Wu K, Fung YK, Ou JH . (1999). Activation of p53 tumor suppressor by hepatitis C virus core protein. Virology 264: 134–141.
Macdonald A, Crowder K, Street A, McCormick C, Harris M . (2004). The hepatitis C virus NS5A protein binds to members of the Src family of tyrosine kinases and regulates kinase activity. J Gen Virol 85: 721–729.
Machida K, Liu JC, McNamara G, Levine A, Duan L, Lai MM . (2009a). Hepatitis C virus causes uncoupling of mitotic checkpoint and chromosomal polyploidy through the Rb pathway. J Virol 83: 12590–12600.
Machida K, Tsukamoto H, Mkrtchyan H, Duan L, Dynnyk A, Liu HM et al. (2009b). Toll-like receptor 4 mediates synergism between alcohol and HCV in hepatic oncogenesis involving stem cell marker Nanog. Proc Natl Acad Sci USA 106: 1548–1553.
Majumder M, Ghosh AK, Steele R, Ray R, Ray RB . (2001). Hepatitis C virus NS5A physically associates with p53 and regulates p21/waf1 gene expression in a p53-dependent manner. J Virol 75: 1401–1407.
Majumder M, Steele R, Ghosh AK, Zhou XY, Thornburg L, Ray R et al. (2003). Expression of hepatitis C virus non-structural 5A protein in the liver of transgenic mice. FEBS Lett 555: 528–532.
Mamiya N, Worman HJ . (1999). Hepatitis C virus core protein binds to a DEAD box RNA helicase. J Biol Chem 274: 15751–15756.
Marukian S, Jones CT, Andrus L, Evans MJ, Ritola KD, Charles ED et al. (2008). Cell culture-produced hepatitis C virus does not infect peripheral blood mononuclear cells. Hepatology 48: 1843–1850.
Masuzaki R, Yoshida H, Tateishi R, Shiina S, Omata M . (2008). Hepatocellular carcinoma in viral hepatitis: improving standard therapy. Best Pract Res Clin Gastroenterol 22: 1137–1151.
Mateu G, Donis RO, Wakita T, Bukh J, Grakoui A . (2008). Intragenotypic JFH1 based recombinant hepatitis C virus produces high levels of infectious particles but causes increased cell death. Virology 376: 397–407.
Mazzocca A, Sciammetta SC, Carloni V, Cosmi L, Annunziato F, Harada T et al. (2005). Binding of hepatitis C virus envelope protein E2 to CD81 up-regulates matrix metalloproteinase-2 in human hepatic stellate cells. J Biol Chem 280: 11329–11339.
McGivern DR, Villanueva RA, Chinnaswamy S, Kao CC, Lemon SM . (2009). Impaired replication of hepatitis C virus containing mutations in a conserved NS5B retinoblastoma protein-binding motif. J Virol 83: 7422–7433.
McHutchison JG, Fried MW . (2003). Current therapy for hepatitis C: pegylated interferon and ribavirin. Clin Liver Dis 7: 149–161.
Mercer DF, Schiller DE, Elliott JF, Douglas DN, Hao C, Rinfret A et al. (2001). Hepatitis C virus replication in mice with chimeric human livers. Nat Med 7: 927–933.
Meurs EF, Galabru J, Barber GN, Katze MG, Hovanessian AG . (1993). Tumor suppressor function of the interferon-induced double-stranded RNA-activated protein kinase. Proc Natl Acad Sci USA 90: 232–236.
Meylan E, Curran J, Hofmann K, Moradpour D, Binder M, Bartenschlager R et al. (2005). Cardif is an adaptor protein in the RIG-I antiviral pathway and is targeted by hepatitis C virus. Nature 437: 1167–1172.
Milward A, Mankouri J, Harris M . (2010). Hepatitis C virus NS5A protein interacts with beta-catenin and stimulates its transcriptional activity in a phosphoinositide-3 kinase-dependent fashion. J Gen Virol 91: 373–381.
Moradpour D, Penin F, Rice CM . (2007). Replication of hepatitis C virus. Nat Rev Microbiol 5: 453–463.
Moriya K, Fujie H, Shintani Y, Yotsuyanagi H, Tsutsumi T, Ishibashi K et al. (1998). The core protein of hepatitis C virus induces hepatocellular carcinoma in transgenic mice. Nat Med 4: 1065–1067.
Moriya K, Nakagawa K, Santa T, Shintani Y, Fujie H, Miyoshi H et al. (2001). Oxidative stress in the absence of inflammation in a mouse model for hepatitis C virus-associated hepatocarcinogenesis. Cancer Res 61: 4365–4370.
Moriya K, Yotsuyanagi H, Shintani Y, Fujie H, Ishibashi K, Matsuura Y et al. (1997). Hepatitis C virus core protein induces hepatic steatosis in transgenic mice. J Gen Virol 78: 1527–1531.
Munakata T, Liang Y, Kim S, McGivern DR, Huibregtse JM, Nomoto A et al. (2007). Hepatitis C virus induces E6AP-dependent degradation of the retinoblastoma protein. PLoS Pathog 3: 1335–1347.
Munakata T, Nakamura M, Liang Y, Li K, Lemon SM . (2005). Down-regulation of the retinoblastoma tumor suppressor by the hepatitis C virus NS5B RNA-dependent RNA polymerase. Proc Natl Acad Sci USA 102: 18159–18164.
Naas T, Ghorbani M, Alvarez-Maya I, Lapner M, Kothary R, De Repentigny Y et al. (2005). Characterization of liver histopathology in a transgenic mouse model expressing genotype 1a hepatitis C virus core and envelope proteins 1 and 2. J Gen Virol 86: 2185–2196.
Nakamoto Y, Guidotti LG, Kuhlen CV, Fowler P, Chisari FV . (1998). Immune pathogenesis of hepatocellular carcinoma. J ExpMed 188: 341–350.
Neumann AU, Lam NP, Dahari H, Gretch DR, Wiley TE, Layden TJ et al. (1998). Hepatitis C viral dynamics in vivo and the antiviral efficacy of interferon-alpha therapy. Science 282: 103–107.
Nomura-Takigawa Y, Nagano-Fujii M, Deng L, Kitazawa S, Ishido S, Sada K et al. (2006). Non-structural protein 4A of Hepatitis C virus accumulates on mitochondria and renders the cells prone to undergoing mitochondria-mediated apoptosis. J Gen Virol 87: 1935–1945.
Norman KL, Sarnow P . (2010). Modulation of hepatitis C virus RNA abundance and the isoprenoid biosynthesis pathway by microRNA miR-122 involves distinct mechanisms. J Virol 84: 666–670.
Okuda M, Li K, Beard MR, Showalter LA, Scholle F, Lemon SM et al. (2002). Mitochondrial injury, oxidative stress, and antioxidant gene expression are induced by hepatitis C virus core protein. Gastroenterology 122: 366–375.
Oshiumi H, Sakai K, Matsumoto M, Seya T . (2010). DEAD/H BOX 3 (DDX3) helicase binds the RIG-I adaptor IPS-1 to up-regulate IFN-beta-inducing potential. Eur J Immunol 40: 940–948.
Otsuka M, Kato N, Lan K, Yoshida H, Kato J, Goto T et al. (2000). Hepatitis C virus core protein enhances p53 function through augmentation of DNA binding affinity and transcriptional ability. J Biol Chem 275: 34122–34130.
Owsianka AM, Patel AH . (1999). Hepatitis C virus core protein interacts with a human DEAD box protein DDX3. Virology 257: 330–340.
Park CY, Choi SH, Kang SM, Kang JI, Ahn BY, Kim H et al. (2009). Nonstructural 5A protein activates beta-catenin signaling cascades: implication of hepatitis C virus-induced liver pathogenesis. J Hepatol 51: 853–864.
Pasquinelli C, Shoenberger JM, Chung J, Chang KM, Guidotti LG, Selby M et al. (1997). Hepatitis C virus core and E2 protein expression in transgenic mice. Hepatology 25: 719–727.
Pavio N, Battaglia S, Boucreux D, Arnulf B, Sobesky R, Hermine O et al. (2005). Hepatitis C virus core variants isolated from liver tumor but not from adjacent non-tumor tissue interact with Smad3 and inhibit the TGF-beta pathway. Oncogene 24: 6119–6132.
Perz JF, Alter MJ . (2006). The coming wave of HCV-related liver disease: dilemmas and challenges. J Hepatol 44: 441–443.
Pineau P, Volinia S, McJunkin K, Marchio A, Battiston C, Terris B et al. (2010). miR-221 overexpression contributes to liver tumorigenesis. Proc Natl Acad Sci USA 107: 264–269.
Prikhod'ko EA, Prikhod'ko GG, Siegel RM, Thompson P, Major ME, Cohen JI . (2004). The NS3 protein of hepatitis C virus induces caspase-8-mediated apoptosis independent of its protease or helicase activities. Virology 329: 53–67.
Qadri I, Iwahashi M, Simon F . (2002). Hepatitis C virus NS5A protein binds TBP and p53, inhibiting their DNA binding and p53 interactions with TBP and ERCC3. Biochim Biophys Acta 1592: 193–204.
Quinkert D, Bartenschlager R, Lohmann V . (2005). Quantitative analysis of the hepatitis C virus replication complex. J Virol 79: 13594–13605.
Randall G, Panis M, Cooper JD, Tellinghuisen TL, Sukhodolets KE, Pfeffer S et al. (2007). Cellular cofactors affecting hepatitis C virus infection and replication. Proc Natl Acad Sci USA 104: 12884–12889.
Ray RB, Steele R, Meyer K, Ray R . (1998). Hepatitis C virus core protein represses p21WAF1/Cip1/Sid1 promoter activity. Gene 208: 331–336.
Sacco R, Tsutsumi T, Suzuki R, Otsuka M, Aizaki H, Sakamoto S et al. (2003). Antiapoptotic regulation by hepatitis C virus core protein through up-regulation of inhibitor of caspase-activated DNase. Virology 317: 24–35.
Saito T, Owen DM, Jiang F, Marcotrigiano J, Gale Jr M . (2008). Innate immunity induced by composition-dependent RIG-I recognition of hepatitis C virus RNA. Nature 454: 523–527.
Sato Y, Kato J, Takimoto R, Takada K, Kawano Y, Miyanishi K et al. (2006). Hepatitis C virus core protein promotes proliferation of human hepatoma cells through enhancement of transforming growth factor alpha expression via activation of nuclear factor-kappaB. Gut 55: 1801–1808.
Schroder M, Baran M, Bowie AG . (2008). Viral targeting of DEAD box protein 3 reveals its role in TBK1/IKKepsilon-mediated IRF activation. Embo J 27: 2147–2157.
Shackel NA, McGuinness PH, Abbott CA, Gorrell MD, McCaughan GW . (2002). Insights into the pathobiology of hepatitis C virus-associated cirrhosis: analysis of intrahepatic differential gene expression. Am J Pathol 160: 641–654.
Shih JW, Tsai TY, Chao CH, Wu Lee YH . (2008). Candidate tumor suppressor DDX3 RNA helicase specifically represses cap-dependent translation by acting as an eIF4E inhibitory protein. Oncogene 27: 700–714.
Shimakami T, Lanford RE, Lemon SM . (2009). Hepatitis C: recent successes and continuing challenges in the development of improved treatment modalities. Curr Opin Pharmacol 9: 537–544.
Shimizu YK, Igarashi H, Kiyohara T, Shapiro M, Wong DC, Purcell RH et al. (1998). Infection of a chimpanzee with hepatitis C virus grown in cell culture. J Gen Virol 79: 1383–1386.
Street A, Macdonald A, Crowder K, Harris M . (2004). The Hepatitis C virus NS5A protein activates a phosphoinositide 3-kinase-dependent survival signaling cascade. J Biol Chem 279: 12232–12241.
Street A, Macdonald A, McCormick C, Harris M . (2005). Hepatitis C virus NS5A-mediated activation of phosphoinositide 3-kinase results in stabilization of cellular beta-catenin and stimulation of beta-catenin-responsive transcription. J Virol 79: 5006–5016.
Sun B, Karin M . (2008). NF-kappaB signaling, liver disease and hepatoprotective agents. Oncogene 27: 6228–6244.
Suzuki HI, Yamagata K, Sugimoto K, Iwamoto T, Kato S, Miyazono K . (2009). Modulation of microRNA processing by p53. Nature 460: 529–533.
Takahashi K, Asabe S, Wieland S, Garaigorta U, Gastaminza P, Isogawa M et al. (2010). Plasmacytoid dendritic cells sense hepatitis C virus-infected cells, produce interferon, and inhibit infection. Proc Natl Acad Sci USA 107: 7431–7436.
Takahashi Y, Rayman JB, Dynlacht BD . (2000). Analysis of promoter binding by the E2F and pRB families in vivo: distinct E2F proteins mediate activation and repression. Genes Dev 14: 804–816.
Tan SL, Nakao H, He Y, Vijaysri S, Neddermann P, Jacobs BL et al. (1999). NS5A, a nonstructural protein of hepatitis C virus, binds growth factor receptor-bound protein 2 adaptor protein in a Src homology 3 domain/ligand-dependent manner and perturbs mitogenic signaling. Proc Natl Acad Sci USA 96: 5533–5538.
Tanaka M, Nagano-Fujii M, Deng L, Ishido S, Sada K, Hotta H . (2006). Single-point mutations of hepatitis C virus NS3 that impair p53 interaction and anti-apoptotic activity of NS3. Bio Biophys Res Comms 340: 792–799.
Tanaka Y, Hanada K, Mizokami M, Yeo AE, Shih JW, Gojobori T et al. (2002). Inaugural Article: a comparison of the molecular clock of hepatitis C virus in the United States and Japan predicts that hepatocellular carcinoma incidence in the United States will increase over the next two decades. Proc Natl Acad Sci USA 99: 15584–15589.
Taura M, Eguma A, Suico MA, Shuto T, Koga T, Komatsu K et al. (2008). p53 regulates Toll-like receptor 3 expression and function in human epithelial cell lines. Mol Cell Biol 28: 6557–6567.
Teufel A, Weinmann A, Centner C, Piendl A, Lohse AW, Galle PR et al. (2009). Hepatocellular carcinoma in patients with autoimmune hepatitis. World J Gastroenterol 15: 578–582.
Thenappan A, Li Y, Kitisin K, Rashid A, Shetty K, Johnson L et al. (2010). Role of transforming growth factor beta signaling and expansion of progenitor cells in regenerating liver. Hepatology 51: 1373–1382.
Thorgeirsson SS, Grisham JW . (2002). Molecular pathogenesis of human hepatocellular carcinoma. Nat Genet 31: 339–346.
Umemura T, Ichijo T, Yoshizawa K, Tanaka E, Kiyosawa K . (2009). Epidemiology of hepatocellular carcinoma in Japan. J Gastroenterol 44 (Suppl 19): 102–107.
Ura S, Honda M, Yamashita T, Ueda T, Takatori H, Nishino R et al. (2009). Differential microRNA expression between hepatitis B and hepatitis C leading disease progression to hepatocellular carcinoma. Hepatology 49: 1098–1112.
Varnholt H, Drebber U, Schulze F, Wedemeyer I, Schirmacher P, Dienes HP et al. (2008). MicroRNA gene expression profile of hepatitis C virus-associated hepatocellular carcinoma. Hepatology 47: 1223–1232.
Wakita T, Pietschmann T, Kato T, Date T, Miyamoto M, Zhao Z et al. (2005). Production of infectious hepatitis C virus in tissue culture from a cloned viral genome. Nat Med 11: 791–796.
Walters KA, Syder AJ, Lederer SL, Diamond DL, Paeper B, Rice CM et al. (2009). Genomic analysis reveals a potential role for cell cycle perturbation in HCV-mediated apoptosis of cultured hepatocytes. PLoS Pathogens 5: e1000269.
Wang AG, Lee DS, Moon HB, Kim JM, Cho KH, Choi SH et al. (2009a). Non-structural 5A protein of hepatitis C virus induces a range of liver pathology in transgenic mice. J Pathol 219: 253–262.
Wang N, Liang Y, Devaraj S, Wang J, Lemon SM, Li K . (2009b). Toll-like receptor 3 mediates establishment of an antiviral state against hepatitis C virus in hepatoma cells. J Virol 83: 9824–9834.
Waris G, Livolsi A, Imbert V, Peyron JF, Siddiqui A . (2003). Hepatitis C virus NS5A and subgenomic replicon activate NF-kappaB via tyrosine phosphorylation of IkappaBalpha and its degradation by calpain protease. J Biol Chem 278: 40778–40787.
Waris G, Tardif KD, Siddiqui A . (2002). Endoplasmic reticulum (ER) stress: hepatitis C virus induces an ER-nucleus signal transduction pathway and activates NF-kappaB and STAT-3. Biochem Pharmacol 64: 1425–1430.
Waris G, Turkson J, Hassanein T, Siddiqui A . (2005). Hepatitis C virus (HCV) constitutively activates STAT-3 via oxidative stress: role of STAT-3 in HCV replication. J Virol 79: 1569–1580.
Whittaker S, Marais R, Zhu AX . (2010). The role of signaling pathways in the development and treatment of hepatocellular carcinoma. Oncogene 29: 4989–5005.
Whyte P, Buchkovich KJ, Horowitz JM, Friend SH, Raybuck M, Weinberg RA et al. (1988). Association between an oncogene and an anti-oncogene: the adenovirus E1A proteins bind to the retinoblastoma gene product. Nature 334: 124–129.
Yeh MM, Daniel HD, Torbenson M . (2010). Hepatitis C-associated hepatocellular carcinomas in non-cirrhotic livers. Mod Pathol 23: 276–283.
Yeoman AD, Al-Chalabi T, Karani JB, Quaglia A, Devlin J, Mieli-Vergani G et al. (2008). Evaluation of risk factors in the development of hepatocellular carcinoma in autoimmune hepatitis: implications for follow-up and screening. Hepatology 48: 863–870.
Yi M, Villanueva RA, Thomas DL, Wakita T, Lemon SM . (2006). Production of infectious genotype 1a hepatitis C virus (Hutchinson strain) in cultured human hepatoma cells. Proc Natl Acad Sci USA 103: 2310–2315.
Yoshizawa H . (2002). Hepatocellular carcinoma associated with hepatitis C virus infection in Japan: projection to other countries in the foreseeable future. Oncology 62 (Suppl 1): 8–17.
You LR, Chen CM, Yeh TS, Tsai TY, Mai RT, Lin CH et al. (1999). Hepatitis C virus core protein interacts with cellular putative RNA helicase. J Virol 73: 2841–2853.
Zhu N, Khoshnan A, Schneider R, Matsumoto M, Dennert G, Ware C et al. (1998). Hepatitis C virus core protein binds to the cytoplasmic domain of tumor necrosis factor (TNF) receptor 1 and enhances TNF-induced apoptosis. J Virol 72: 3691–3697.
Acknowledgements
This work was supported in part by the University Cancer Research Fund and grants from the National Institutes of Health: P20-CA127004 and R21-AI081058.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
McGivern, D., Lemon, S. Virus-specific mechanisms of carcinogenesis in hepatitis C virus associated liver cancer. Oncogene 30, 1969–1983 (2011). https://doi.org/10.1038/onc.2010.594
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2010.594
Keywords
This article is cited by
-
An overview of mouse models of hepatocellular carcinoma
Infectious Agents and Cancer (2023)
-
Association of MTHFR and TYMS gene polymorphisms with the susceptibility to HCC in Egyptian HCV cirrhotic patients
Clinical and Experimental Medicine (2022)
-
Effects of hepatitis C virus core protein and nonstructural protein 4B on the Wnt/β-catenin pathway
BMC Microbiology (2017)
-
Immunotherapy against cancer-related viruses
Cell Research (2017)
-
Hepatitis C virus drives the pathogenesis of hepatocellular carcinoma: from immune evasion to carcinogenesis
Clinical & Translational Immunology (2016)