Summary
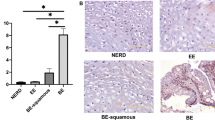
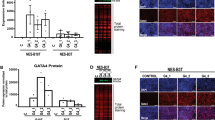
The protein expression patterns of normal, metaplastic and malignant oesophageal tissues were analysed by two-dimensional polyacrylamide gel electrophoresis (2D-PAGE) to identify changes associated with Barrett’s metaplasia and transformation to oesophageal adenocarcinoma. Heat-shock protein 27 (Hsp27), a small heat-shock protein which is protective against cytotoxic stresses, was abundant in normal oesophagus. However, Hsp27 expression was markedly lower in Barrett’s metaplasia and oesophageal adenocarcinomas. This was confirmed by immunohistochemical analysis. Hsp27 protein was most highly expressed in the upper layers of squamous epithelium and exhibited a pattern of expression that corresponded with the degree of squamous maturation. Northern and Southern analysis demonstrated Hsp27 to be regulated at the level of mRNA transcription or abundance. Normal oesophageal tissues were examined for gender differences in Hsp27 expression. Women expressed fourfold higher levels of Hsp27 mRNA, however, this difference was not appreciable in protein expression. Hsp27 protein was inducible by heat shock in Barrett’s adenocarcinoma cell lines and an immortalized oesophageal epithelial cell line (HET-1A), but not by oestradiol. These results demonstrate abundant constitutive expression of the stress-response protein Hsp27 in the normal oesophagus, and suggest that low-level expression in Barrett’s metaplasia may be one factor which may influence susceptibility to oesophageal adenocarcinoma development.
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Al-Kasspooles, M., Moore, J. H., Orringer, M. B. & Beer, D. G. (1993). Amplification and overexpression of the EGFR and erbB-2 genes in human esophageal adenocarcinomas. Int J Cancer 54: 213–219.
Ames, B. N., Shigenaga, M. K. & Hagen, T. M. (1993). Oxidants, antioxidants, and the degenerative diseases of aging. Proc Natl Acad Sci, USA 90: 7915–7922.
Arrigo, A-P & Welch, W. J. (1987). Characterization and purification of the small 28,000-Dalton mammalian heat shock protein. J Biol Chem 262: 15359–15369.
Barrett, N. R. (1950). Chronic peptic ulcer of the oesophagus and ‘oesophagitis’. Br J Surg 38: 175–182.
Barrett, N. R. (1957). The lower esophagus lined by columnar epithelium. Surgery 41: 881–894.
Blot, W. J., Devesa, S. S., Kneller, R. W. & Fraumeni, J. F. (1991). Rising incidence of adenocarcinoma of the esophagus and gastric cardia. JAMA 265: 1287–1289.
Bremner, C. G., Lynch, V. P. & Ellis, F. H. (1970). Barrett’s esophagus: congenital or acquired? An experimental study of esophageal mucosal regeneration in the dog. Surgery 68: 209–216.
Cameron, A. J., Ott, B. J. & Payne, W. S. (1985). The incidence of adenocarcinoma in columnar-lined (Barrett’s) esophagus. N Engl J Med 313: 857–859.
Ciocca, R. D., Adams, D. J., Edwards, D. P., Bjercke, R. J. & McGuire, W. L. (1983). Distribution of an estrogen-induced protein with a molecular weight of 24,000 in normal and malignant human tissues and cells. Cancer Res 43: 1204–1210.
Dressler, L. G., Ramzy, I., Sledge, G. W. & McGuire, W. L. (1986). A new marker of maturation in the cervix: the estrogen-regulated 24k protein. Obstet Gynecol 68: 825–831.
Dunn, D. K., Whelan, R. D. H., Hill, B. & King, R. J. B. (1993). Relationship of HSP27 and oestrogen receptor in hormone sensitive and insensitive cell lines. J Steroid Biochem Mol Biol 46: 469–479.
Edwards, D. P., Adams, D. J., Savage, N. & McGuire, W. L. (1980). Estrogen induced synthesis of specific proteins in human breast cancer cells. Biochem Biophys Res Commun 93: 804–812.
Fiers, W. (1991). Tumor necrosis factor: characterization at the molecular, cellular and in vivo level. FEBS Lett 285: 199–212.
Fuqua, S. A. W., Blum-Salingaros, M. & McGuire, W. L. (1989). Induction of the estrogen-regulated ‘24K’ protein by heat shock. Cancer Res 49: 4126–4129.
Goldman, M. C. & Beckman, R. C. (1960). Barrett syndrome: case report with discussion about concepts of pathogenesis. Gastroenterology 39: 104–110.
Hamilton, S. R. (1992). Oesophagitis. In Pathology of the Gastrointestinal Tract, Ming S-C and Goldman H (eds), pp. 381–438, WB Saunders: Philadelphia
Hanash, S. M., Baier, L. J., McCurry, L. & Schwartz, S. A. (1986). Lineage-related polypeptide markers in acute lymphoblastic leukemia detected by two-dimensional gel electrophoresis. Proc Natl Acad Sci USA 83: 807–811.
Hanash, S. M., Kuick, R., Nichols, D. & Stoolman, L. (1988). Quantitative analysis of a new marker for common acute lymphoblastic leukemia detected by two-dimensional electrophoresis. Dis Markers 6: 209–220.
Hanson, L. A., Nuzum, E. O., Jones, B. C., Malkinson, A. M. & Beer, D. G. (1991). Expression of the glucocorticoid receptor and K-ras genes in urethan-induced mouse lung tumors and transformed cell lines. Exp Lung Res 17: 371–387.
Hesketh, P. J., Clapp, R. W., Doos, W. G. & Spechler, S. J. (1989). The increasing frequency of adenocarcinoma of the esophagus. Cancer 64: 526–530.
Hickey, E., Brandon, S. E., Sadis, S., Smale, G. & Weber, L. A. (1986). Molecular cloning of sequences encoding the human heat-shock proteins and their expression during hyperthermia. Gene 43: 147–154.
Hinder, R. A. & Stein, H. J. (1991). Oxygen-derived free radicals. Arch Surg 126: 104–105.
Hout, J., Roy, G., Lambert, H., Chretien, P. & Landry, J. (1991). Increased survival after treatments with anticancer agents of Chinese hamster cells expressing the human Mr 27,000 heat shock protein. Cancer Res 51: 5245–5252.
Hout, J., Houle, F., Spitz, D. R. & Landry, J. (1996). Hsp27 phosphorylation-mediated resistance against actin fragmentation and cell death induced by oxidative stress. Cancer Res 56: 273–279.
Kuick, R. D., Hanash, S. M., Chu, E. H. Y. & Strahler, J. R. (1987). A comparison of some adjustment techniques for use with quantitative spot data from two-dimensional gels. Electrophoresis 8: 199–204.
Landry, J., Chretien, P., Lambert, H., Hickey, E. & Weber, L. A. (1989). Heat shock resistance conferred by expression of the human hsp27 gene in rodent cells. J Cell Biol 109: 7–15.
Landry, J., Chretien, P., Laszlo, A. & Lambert, H. (1991). Phosphorylation of Hsp27 during development and decay of thermotolerance in Chinese hamster cells. J Cell Physiol 147: 93–101.
Landry, J., Lambert, H., Zhou, M., Lavoie, J. N., Hickey, E., Weber, L. A. & Anderson, C. W. (1992). Human Hsp27 is phosphorylated at serines 78 and 82 by heat shock and mitogen-activated kinases that recognize the same amino acid motif as S6 kinase II. J Biol Chem 267: 794–803.
Lavoie, J. N., Lambert, H., Hickey, E., Weber, L. A. & Landry, J. (1995). Modulation of cellular thermoresistance and actin filament stability accompanies phosphorylation-induced changes in the oligomeric structure of heat shock protein 27. Mol Cell Biol 15: 505–516.
McBride, T. J., Preston, B. D. & Loeb, L. A. (1991). Mutagenic spectrum resulting from DNA damage by oxygen radicals. Biochemistry 30: 207–213.
Mehlen, P., Mehlen, A., Guillet, D., Preville, X. & Arrigo, A-P (1995a). Tumor necrosis factor-α induces changes in the phosphorylation, cellular localization, and oligomerization of human hsp27, a stress protein that confers cellular resistance to this cytokine. J Cell Biochem 58: 248–259.
Mehlen, P., Preville, X., Chareyron, P., Briolay, J., Klemenz, R. & Arrigo, A-P (1995b). Constitutive expression of human hsp27, Drosophila hsp27, or human aB-crystallin confers resistance to TNF- and oxidative stress-induced cytotoxicity in stably transfected murine L929 fibroblasts. J Immunol 154: 363–374.
Mehlen, P., Kretz-Remy, C., Preville, X. & Arrigo, A-P (1996). Human hsp27, Drosophila hsp27 and human αB-crystallin expression-mediated increase in glutathione is essential for the protective activity of these proteins against TNFα-induced cell death. EMBO J 15: 2695–2706.
Moscow, J. A., Fairchild, C. R., Madden, M. J., Ransom, D. T., Wieand, H. S., O’Brien, E. E., Poplack, D. G., Cossman, J., Myers, C. E. & Cowan, K. H. (1989). Expression of anionic glutathione-S-transferase and p-glycoprotein genes in human tissues and tumors. Cancer Res 49: 1422–1428.
Naef, A. P., Savary, M. & Ozzello, L. (1975). Columnar-lined lower esophagus: an acquired lesion with malignant predisposition. J Thorac Cardiovasc Surg 70: 826–835.
Olyaee, M., Sontag, S., Salman, W., Schnell, T., Mobarhan, S., Eiznhamer, D. & Keshavarzian, A. (1995). Mucosal reactive oxygen species production in oesophagitis and Barrett’s oesophagus. Gut 37: 168–173.
Rachwal, W. J., Bongiorno, P. F., Orringer, M. B., Whyte, R. I., Ethier, S. P. & Beer, D. G. (1995). Expression and activation of erbB-2 and epidermal growth factor receptor in lung adenocarcinomas. Br J Cancer 72: 56–64.
Reed, P. I. & Johnson, B. J. (1993). The changing incidence of oesophageal cancer. Endoscopy 25: 606–608.
Richards, E. H., Hickey, E., Weber, L. & Masters, J. R. W. (1996). Effect of overexpression of the small heat shock protein HSP27 on the heat and drug sensitivities of human testis tumor cells. Cancer Res 56: 2446–2451.
Schnell, T., Sontag, S., Wanner, J., Chintam, R., Chejfec, G., O’Connell, S. & Moroni, B. (1985). Endoscopic screening for Barrett’s esophagus (BE), esophageal adenocarcinoma (AdCa) and other mucosal changes in ambulatory subjects with symptomatic gastroesophageal reflux (GER). Gastroenterology 88: 1576
Skinner, D. B., Walther, B. C., Riddell, R. H., Schmidt, H., Iascone, C. & Demeester, T. R. (1983). Barrett’s esophagus. Comparison of benign and malignant cases. Ann Surg 198: 554–566.
Spechler, S. J., Robbins, A. H., Rubins, H. B., Vincent, M. E., Heeren, T., Doos, W. G., Colton, T. & Schimmel, E. M. (1984). Adenocarcinoma and Barrett’s esophagus: an overrated risk?. Gastroenterology 87: 927–933.
Stoner, G. D., Kaighn, M. E., Reddel, R. R., Resau, J. H., Bowman, D., Naito, Z., Matsukura, N., You, M., Galati, A. J. & Harris, C. C. (1991). Establishment and characterization of SV40 T-antigen immortalized human esophageal epithelia cells. Cancer Res 51: 365–371.
Strahler, J. R., Kuick, R., Eckerskorn, C., Lottspeich, F., Richardson, B. C., Fox, D. A., Stoolman, L. M., Hanson, C. A., Nichols, D., Tueche, H. J. & Hanash, S. M. (1990). Identification of two related markers for common acute lymphoblastic leukemia as heat shock proteins. J Clin Invest 85: 200–207.
Trautinger, F., Kindas-Mugge, I., Dekrout, B., Knobler, R. M. & Metze, D. (1995). Expression of the 27-kDa heat shock protein in human epidermis and in epidermal neoplasms: an immunohistological study. Br J Dermatol 133: 194–202.
Trush, M. A. & Kensler, T. W. (1991). An overview of the relationship between oxidative stress and chemical carcinogenesis. Free Radical Biol Med 10: 201–209.
Van Der Veen, A. H., Dees, J., Blankensteijn, J. D. & Van Blankenstein, M. (1989). Adenocarcinoma in Barrett’s oesophagus: an overrated risk. Gut 30: 14–18.
Welch, W. J. (1985). Phorbol ester, calcium ionophore, or serum added to quiescent rat embryo fibroblast cells all result in the elevated phosphorylation of two 28,000-Dalton mammalian stress proteins. J Biol Chem 260: 3058–3062.
Wetscher, G. J., Hinder, P. R., Bagchi, D., Perdikis, G., Redmond, E. J., Glaser, K., Adrian, T. E. & Hinder, R. A. (1995a). Free radical scavengers prevent reflux esophagitis in rats. Dig Dis Sci 40: 1292–1296.
Wetscher, G. J., Hinder, R. A., Bagchi, D., Hinder, P. R., Bagchi, M., Perdikis, G. & McGinn, T. (1995b). Reflux esophagitis in humans is mediated by oxygen-derived free radicals. Am J Surg 170: 552–557.
Wetscher, G. J., Perdikis, G., Kretchmar, D. H., Stinson, R. G., Bagchi, D., Redmond, E. J., Adrian, T. E. & Hinder, R. A. (1995c). Esophagitis in Sprague-Dawley rats is mediated by free radicals. Dig Dis Sci 40: 1297–1305.
Wheater, P. R., Burkitt, H. G. & Daniels, V. G. (1987). Functional Histology: a Text and Colour Atlas, pp. 64–78, Churchill Livingstone: New York
Winters, C., Spurling, T. J., Chobanian, S. J., Curtis, D. J., Esposito, R. L., Hacker, J. F., Johnson, D. A., Cruess, D. F., Cotelingam, J. D., Gurney, M. S. & Cattau, E. L. (1987). Barrett’s esophagus: a prevalent, occult complication of gastroesophageal reflux disease. Gastroenterology 92: 118–124.
Wu, W. & Welsh, M. J. (1996). Expression of the 25-kDa heat-shock protein (HSP27) correlates with resistance to the toxicity of cadmium chloride, mercuric chloride, cis-platinum(II)-diammine dichloride, or sodium arsenite in mouse embryonic stem cells transfected with sense or antisense HSP27 cDNA. Toxicol Appl Pharmacol 141: 330–339.
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Soldes, O., Kuick, R., Thompson II, I. et al. Differential expression of Hsp27 in normal oesophagus, Barrett’s metaplasia and oesophageal adenocarcinomas. Br J Cancer 79, 595–603 (1999). https://doi.org/10.1038/sj.bjc.6690094
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bjc.6690094
Keywords
This article is cited by
-
Galectin-3 expression is prognostic in diffuse type gastric adenocarcinoma, confers aggressive phenotype, and can be targeted by YAP1/BET inhibitors
British Journal of Cancer (2018)
-
Nuclear expression of Gli-1 is predictive of pathologic complete response to chemoradiation in trimodality treated oesophageal cancer patients
British Journal of Cancer (2017)
-
The Functions of Antioxidants and Heat Shock Proteins Are Altered in the Immune Organs of Selenium-Deficient Broiler Chickens
Biological Trace Element Research (2016)
-
Hsp27 (HSPB1) differential expression in normal salivary glands and pleomorphic adenomas and association with an increased Bcl2/Bax ratio
Tumor Biology (2015)
-
A mouse model for triple-negative breast cancer tumor-initiating cells (TNBC-TICs) exhibits similar aggressive phenotype to the human disease
BMC Cancer (2012)